Global NICU Genetic Testing Market Size & Trends Report Segmented by Offering (Instruments, Consumables, Services), Testing Type (WGS, WES, CMA), Indication (Congenital Anomalies, Metabolic Disorders) End-user (Hospitals, Maternity/Specialty Clinics) & Regional Forecast to 2031
The global NICU genetic testing market size is set to witness a growth rate of 12% in the next 5 years. Increasing incidence of genetic and rare disorders in neonates, growing adoption of rapid genomic sequencing (rWGS/rWES), shift toward precision and personalized neonatal care, expanding newborn screening programs globally, increasing awareness among clinicians and parents, and supportive government initiatives and funding are some of the key factors driving the NICU genetic testing market. To learn more about the research report, download a sample report.
Neonatal intensive care unit (NICU) genetic testing refers to the use of cutting-edge genomic and molecular diagnostic technologies to find genetic disorders in critically ill newborns admitted to a NICU. Some of the technologies that are commonly used for this purpose include next-generation sequencing (NGS), whole exome sequencing (WES), and rapid whole-genome sequencing (rWGS), which can identify the genetic basis for unexplained clinical signs, congenital malformations, and potentially life-threatening conditions. The primary purpose of NICU genetic testing is to provide early and accurate diagnoses to facilitate timely clinical decision-making, individualized management strategies, and improved patient outcomes. Genetic testing used in the NICU primarily occurs in emergency/urgent situations and high-risk patients where quick results are necessary to guide therapeutic interventions and decrease the potential for morbidity or mortality of neonates.
Increasing incidence of genetic and rare disorders in neonates to propel market demand
The increasing number of newborns suffering from genetic and rare disorders is strongly contributing to the growth of the NICU genetic testing market. For instance, the global burden of congenital birth defects (CBDs) has increased, reaching approximately 31.64 million children in 2021, representing a 6.68% rise since 1990, with nearly 6% of infants born with a disorder each year (Source: NCBI). As a result, there has been an increase in the need for timely, correct diagnostic solutions in critical care facilities. Many diseases that present in neonates, such as congenital abnormalities like congenital heart defect; metabolic disorders like cystic fibrosis; neurological diseases like cerebral palsy, and syndromic diseases like Down syndrome, among others have an underlying genetic cause. Furthermore, many of these disorders exhibit either non-specific or overlapping clinical presentations; thus, routine diagnostic methods may take longer to diagnose the underlying cause of the illness.
As clinicians become aware of the greater frequency of these types of conditions, they increasingly utilize new diagnostic genetics technologies to find the underlying etiology of the disorder. Since prompt and accurate diagnosis is particularly critical in NICUs, delayed treatment can lead to serious complications, long-term disabilities and deaths. The ability to provide accurate diagnosis through genetic testing allows clinicians to progress from managing based on clinical signs or symptoms to providing specific treatment for individual patients. This ability should result in improved clinical outcomes overall for infants admitted to NICUs. Advances in neonatal healthcare have improved survival rates among premature and critically ill newborns, leading to a growing population of infants requiring specialized medical follow-up. As survival increases, more newborns require genetic evaluation to identify potential genetic conditions that may affect long-term health outcomes and developmental trajectories. The growing number of infants who have survived the initial period of critical illness creates an increased need for diagnosing genetic conditions that may contribute to this increased number and frequency of newborns with genetic conditions and therefore creating an increased demand from NICUs for genetic testing in their respective institutions.
The increased awareness among both healthcare providers and parents about the importance of genetics in neonatal disease is also promoting growth of this market. Healthcare providers are now more likely to offer genetic testing as a primary diagnostic tool for newborn patients that are diagnosed with unexplained symptoms or have a family history of inherited disorders. Also, with the continued growth in the number of genetic conditions that can be diagnosed through traditional genetic methods and the increased volume of genetic research, these results have made genetic testing even more relevant to the healthcare community and provide additional insight into the significance of genetic evaluation of newborns.
In conclusion, as the prevalence of genetic and rare disorders continues to rise among newborns, there is a growing need and value placed on timely and accurate diagnosis through NICU-led genetic testing methods. Therefore, due to the significant impact genetic testing provides to the modern-day practice of neonatal medicine, the demand for genetic testing in NICUs is likely to continue to grow.

To learn more about this report, download the PDF brochure
Growing adoption of rapid genomic sequencing is driving the market growth
Rapid growth in the adoption of rapid genomic sequencing through rapid whole genome sequencing (rWGS) and rapid whole exome sequencing (rWES) is a primary driver of growth for the NICU genetic testing market due to the significant demand for quick and precise diagnoses in infants who are critically ill. In NICU, infants typically arrive with complex, unexplained clinical symptoms that necessitate urgent medical intervention. Traditional methods of diagnosis can take weeks to months for the results to be available before making treatment decisions. Rapid genomic sequencing can generate results between 24 and 72 hours, which is extremely valuable in being able to administer care to an acutely ill infant. With this expedited turnaround time, a clinician can determine the genetic cause of the medical condition, enabling them to provide targeted therapies, oversee appropriate clinical management, and avoid providing unnecessary and/or ineffective treatments to their patient. Early diagnosis using rWGS or rWES is associated with increased survival rates, fewer complications, and shorter lengths of stay in the hospital, thus improving overall outcomes for the patient. Based on this information, the incorporation of rapid sequencing into the routine workflows of hospitals and healthcare systems in the NICU is becoming increasingly prevalent.
Rapid genomic sequencing has become easier to perform, faster, and less complex due to advances in technology. For example, sequencing platform improvements, more automation, and better bioinformatics are producing improved accuracy with shorter turn-around times and less operating complexity. This has led to a larger number of healthcare facilities using rapid genomic sequencing, especially in mature regions where infrastructure and expertise exist.
Another factor that is notable in increasing the adoption of rapid genomic sequencing by hospitals is evidence that supports the utility and cost-effectiveness of rapid genomic sequencing. Numerous studies indicate that getting a genetic diagnosis early can save costs through reduced diagnostic delays and the need for intensive care. The increasing amount of research in this area is influencing healthcare policy regarding reimbursement for services related to rapid genomic sequencing.
Rapid genomic sequencing is being adopted in NICUs as an integral part of the clinical care plan because of growing clinician awareness and advances in precision medicine. As healthcare providers continue moving toward personalized medicine, rWGS and rWES is likely to become cornerstone for providing personalized therapies based on the infant's genetic make-up. In summary, widespread use of rapid genomics is likely to have a profound effect on NICU diagnostics making them quicker, more precise, and more clinically significant resulting in rapid growth of the NICU genetics testing market.
To learn more about this report, download the PDF brochure
Testing Type Segment Outlook
Whole genome sequencing (WGS) represents the largest share of the NICU genetic testing market due to its all-inclusive detection abilities for a broad spectrum of genetic variation, thereby making it indispensable when diagnosing complex and critically ill newborn patients. The accelerating use of rapid testing formats continues to develop WGS’s position as the primary driver of growth in the industry. Likewise, whole exome sequencing (WES) is projected to experience the fastest growth rate throughout the forecast period due to its cost-effective nature, shorter turnaround time, and established clinical utility in the identification of variants that cause disease in the coding regions of the exome, thus becoming the method of choice in both resource-limited and routine clinical settings.
Regional Outlook: North America leads NICU genetic testing market due to advanced healthcare infrastructure while improving healthcare systems and awareness drive growth in APAC
North America held the largest share of the NICU genetic testing market owing to its advanced health care infrastructure, widespread adoption of NGS technologies, presence of several major genomic firms in the area, and favourable reimbursement policies. There are also numerous ongoing investments in precision medicine as well as established systems for providing quality neonatal care throughout North America. In Asia-Pacific, however, the NICU genetic testing market is expected to grow at the fastest rate during the forecast period, supported by improvements to the health care systems, greater public awareness of genetic disorders, increasing birth rates, and stronger government involvement in expanding newborn screening programs and the development of genomic medicine. In addition, increased availability of advanced diagnostics and higher health care expenditures are likely to support continued growth of the NICU genetic testing marketplaces in emerging economies in the APAC region.
Competitive Landscape Analysis
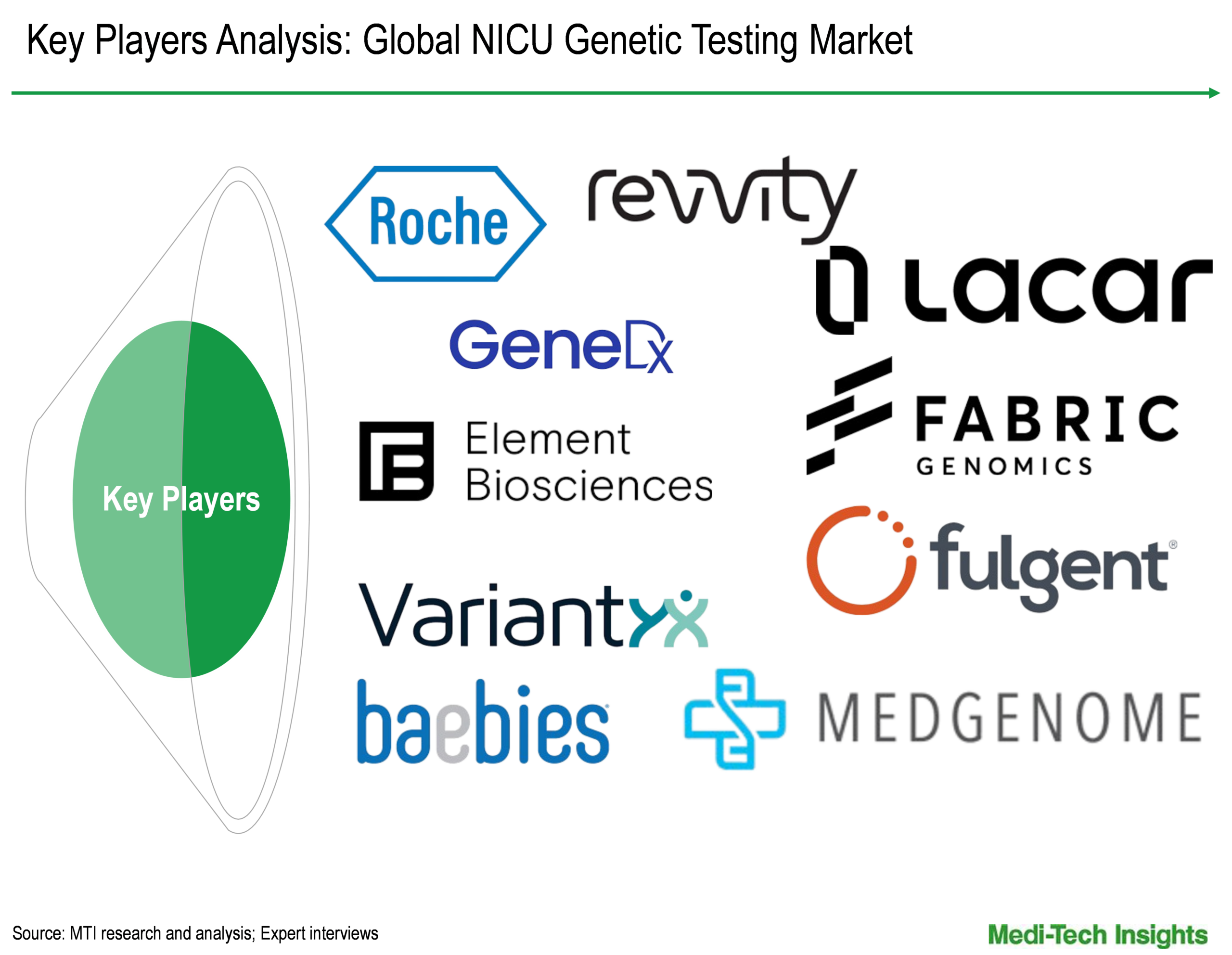
The global NICU genetic testing market is marked by the presence of established and emerging market players such as F. Hoffmann-La Roche Ltd (Switzerland); Revvity (US); GeneDx, LLC (US); Element Biosciences (US); LaCAR MDx (Belgium); Baebies (US); Fulgent Genetics (US); MedGenome (India); Variantyx, Inc. (US); and Fabric Genomics (US), among others.
Get a sample report for competitive landscape analysis
Growth strategies adopted by players to establish their foothold in the market
Players operating in this market are adopting various growth strategies such as new product and service launches; acquisitions; and strategic partnerships, agreements, and collaborations, to garner market share. For instance,
- In May 2025, F. Hoffmann-La Roche Ltd partnered with Broad Clinical Labs to develop and pilot applications of its SBX sequencing technology, focusing on critically ill newborns and trio-based genome sequencing. The collaboration aims to accelerate adoption of ultra-fast, scalable sequencing and establish whole genome sequencing as a routine component of NICU clinical care
- In May 2025, GeneDx, Inc. completed the acquisition of Fabric Genomics to enhance AI-driven genomic analysis and expand global diagnostic capabilities. The acquisition strengthens NICU genetic testing by enabling faster, scalable, and integrated end-to-end genomic solutions, improving time-to-diagnosis for critically ill newborns
- In February 2025, GeneDx, Inc. announced ultraRapid whole genome sequencing capable of delivering actionable results within 48 hours for critically ill NICU and PICU patients. The solution aims to accelerate diagnosis, reduce NICU stay and unnecessary treatments, and generate healthcare savings of over $15,000 per child while addressing underutilization of genetic testing
Report Scope
|
Report Metric |
Details |
|
Base Year Considered |
2025 |
|
Historical Data |
2024 – 2025 |
|
Forecast Period |
2026 – 2031 |
|
Growth Rate |
12% |
|
Segment Scope |
Offering, Testing Type, Indication, End User |
|
Regional Scope |
|
|
Market Drivers |
|
|
Attractive Opportunities |
|
|
Key Companies Mapped |
F. Hoffmann-La Roche Ltd (Switzerland); Revvity (US); GeneDx, LLC (US); Element Biosciences (US); LaCAR MDx (Belgium); Baebies (US); Fulgent Genetics (US); MedGenome (India); Variantyx, Inc. (US); and Fabric Genomics (US); among others |
|
Report Highlights |
Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Global NICU Genetic Testing Market Segmentation
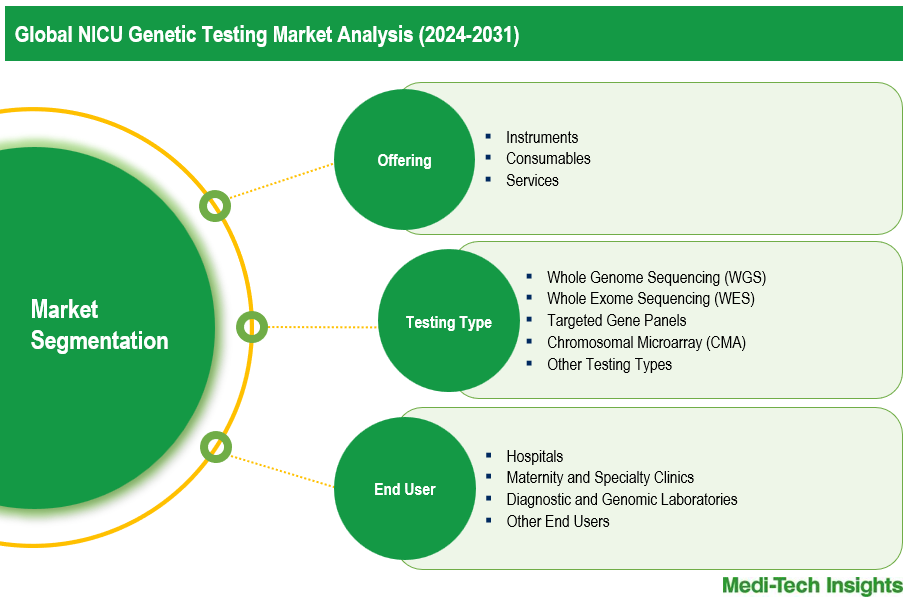
This report by Medi-Tech Insights provides the size of the global NICU genetic testing market at the regional- and country-level from 2024 to 2031. The report further segments the market based on offering, testing type, indication, and end user.
Market Size & Forecast (2024-2031), By Offering, USD Billion
- Instruments
- Consumables
- Services
Market Size & Forecast (2024-2031), By Testing Type, USD Billion
- Whole Genome Sequencing (WGS)
- Whole Exome Sequencing (WES)
- Targeted Gene Panels
- Chromosomal Microarray (CMA)
- Other Testing Types
Market Size & Forecast (2024-2031), By Indication, USD Billion
- Congenital Anomalies
- Neurological Disorders
- Metabolic Disorders
- Rare Diseases
- Other Indications
Market Size & Forecast (2024-2031), By End User, USD Billion
- Hospitals
- Maternity and Specialty Clinics
- Diagnostic and Genomic Laboratories
- Other End Users
Market Size & Forecast (2024-2031), By Region, USD Billion
- North America
- US
- Canada
- Europe
- UK
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Middle East & Africa
- Introduction
- Introduction
- Market Scope
- Market Definition
- Segments Covered
- Regional Segmentation
- Research Timeframe
- Currency Considered
- Study Limitations
- Stakeholders
- List of Abbreviations
- Key Conferences and Events (2026-2027)
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Bottom-Up Approach
- Top-Down Approach
- Market Forecasting
- Executive Summary
- NICU Genetic Testing Market Snapshot (2026-2031)
- Segment Overview
- Regional Snapshot
- Competitive Insights
- Market Overview
- Market Dynamics
- Drivers
- Rising incidence of genetic and rare disorders in neonates
- Growing adoption of rapid genomic sequencing (rWGS / rWES)
- Shift toward precision and personalized neonatal care
- Expanding newborn screening programs globally
- Increasing awareness among clinicians and parents
- Supportive government initiatives and funding
- Restraints
- High cost of genetic testing
- Limited reimbursement coverage
- Complex data interpretation
- Ethical and regulatory concerns
- Opportunities
- Integration of genomics into standard NICU workflows
- Expansion in emerging markets
- AI and bioinformatics advancements
- Growth in rare disease diagnostics ecosystem
- Development of targeted and gene therapies
- Increasing adoption of non-invasive and minimal sample testing
- Key Market Trends
- Shift from reactive to proactive diagnostics
- Expansion of multi-omics approaches
- Growth of centralized genomic labs
- Unmet Market Needs
- Industry Speaks
- Drivers
- Market Dynamics
- Global NICU Genetic Testing Market Size & Forecast (2024-2031), By Offering, USD Billion
- Introduction
- Instruments
- Consumables
- Services
- Global NICU Genetic Testing Market Size & Forecast (2024-2031), By Testing Type, USD Billion
- Introduction
- Whole Genome Sequencing (WGS)
- Whole Exome Sequencing (WES)
- Targeted Gene Panels
- Chromosomal Microarray (CMA)
- Other Testing Types
- Global NICU Genetic Testing Market Size & Forecast (2024-2031), By Indication, USD Billion
- Introduction
- Congenital Anomalies
- Neurological Disorders
- Metabolic Disorders
- Rare Diseases
- Other Indications
- Global NICU Genetic Testing Market Size & Forecast (2024-2031), By End User, USD Billion
- Introduction
- Hospitals
- Maternity and Specialty Clinics
- Diagnostic and Genomic Laboratories
- Other End Users
- Global NICU Genetic Testing Market Size & Forecast (2024-2031), By Region, USD Billion
- Introduction
- North America NICU Genetic Testing Market Size & Forecast (2024-2031), By Country, USD Billion
- US
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Canada
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion))
- US
- Europe NICU Genetic Testing Market Size & Forecast (2024-2031), By Country, USD Billion
- UK
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Germany
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- France
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Italy
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Spain
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Rest of Europe
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- UK
- Asia Pacific (APAC) NICU Genetic Testing Market Size & Forecast (2024-2031), By Country, USD Billion
- China
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Japan
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- India
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Rest of Asia Pacific
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- China
- Latin America (LATAM) NICU Genetic Testing Market Size & Forecast (2024-2031), USD Billion
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Middle East & Africa (MEA) NICU Genetic Testing Market Size & Forecast (2024-2031), USD Billion
- Market Size & Forecast, By Offering, (USD Billion)
- Market Size & Forecast, By Testing Type, (USD Billion)
- Market Size & Forecast, By Indication, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Key Player Comparison
- Segment-wise Player Mapping
- Market Share Analysis (2025)
- Company Categorization Matrix
- Dominants/Leaders
- New Entrants
- Emerging Players
- Innovative Players
- Key Strategies Assessment, By Player (2023-2026)
- New Product and Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Company Profiles*
(Business Overview, Financial Performance**, Products Offered, Recent Developments)
- Hoffmann-La Roche Ltd
- Revvity
- GeneDx, LLC
- Element Biosciences
- LaCAR MDx
- Baebies
- Fulgent Genetics
- MedGenome
- Variantyx, Inc.
- Fabric Genomics
- Other Prominent Players
Note: *Indicative list
**For listed companies
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)

Primary Research
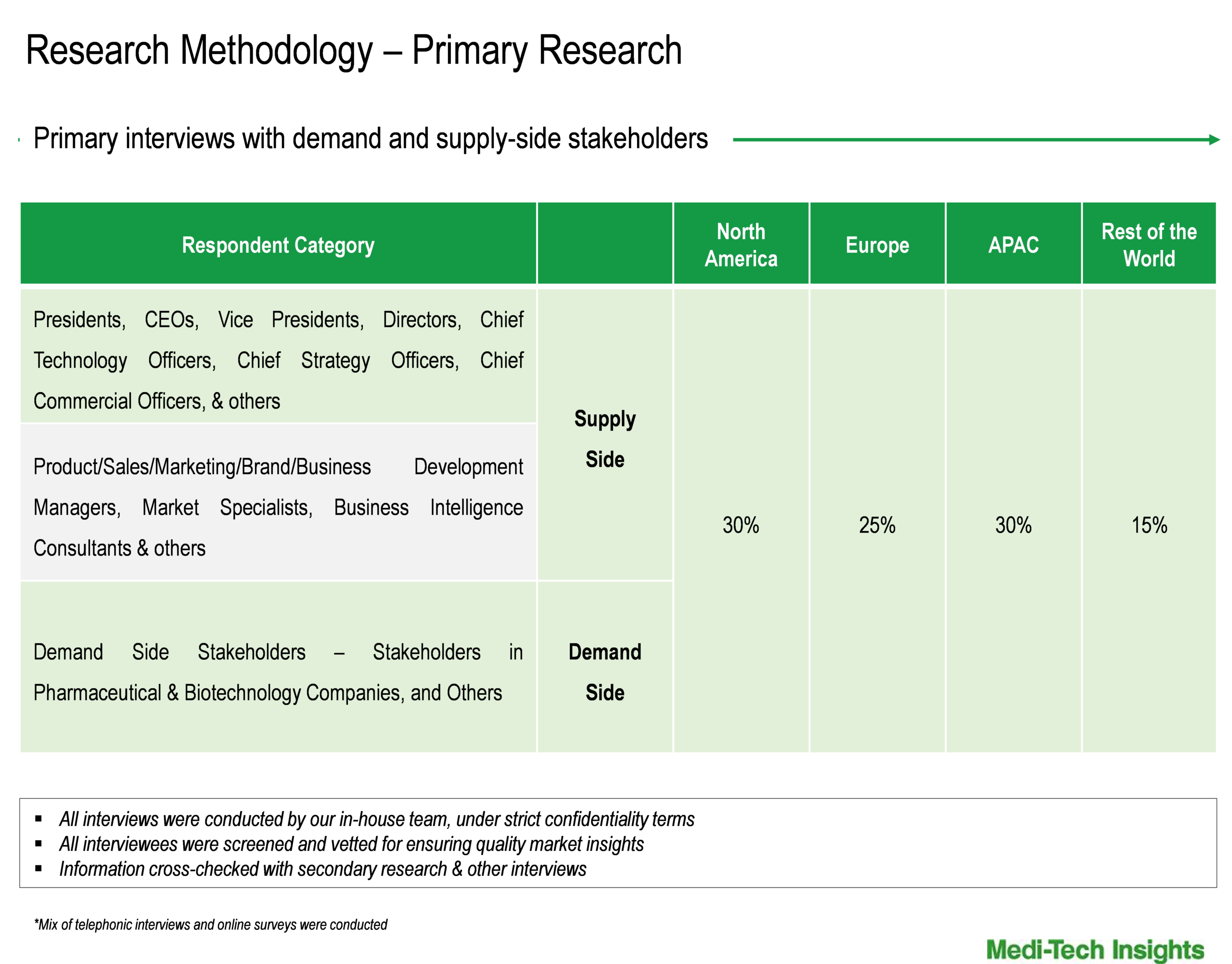
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Hospitals, Maternity and Specialty Clinics, and Others
Breakdown of Primary Interviews
Market Size Estimation
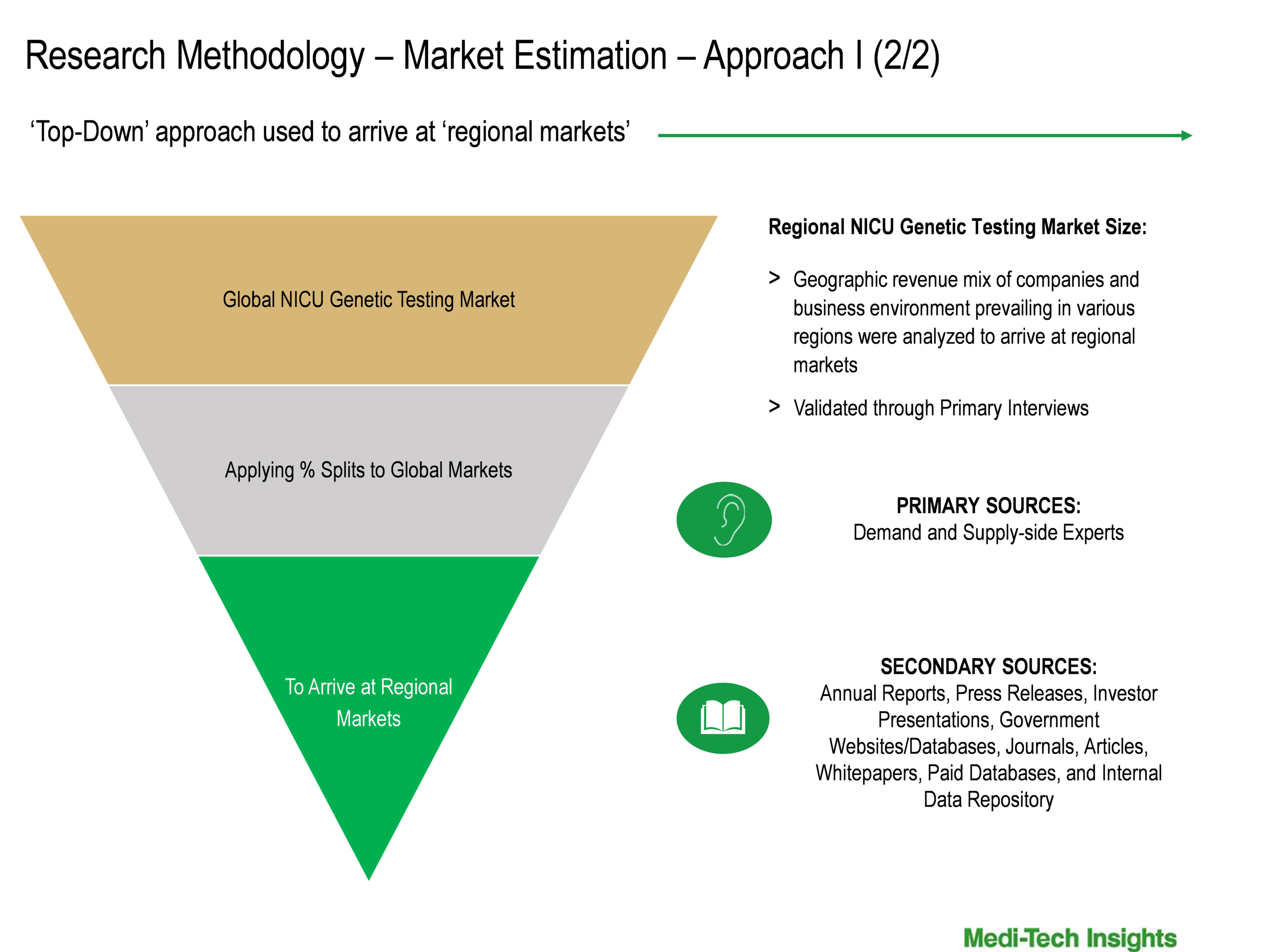
Both ‘Top-Down & Bottom-Up Approaches’ were used to derive market size estimates and forecasts
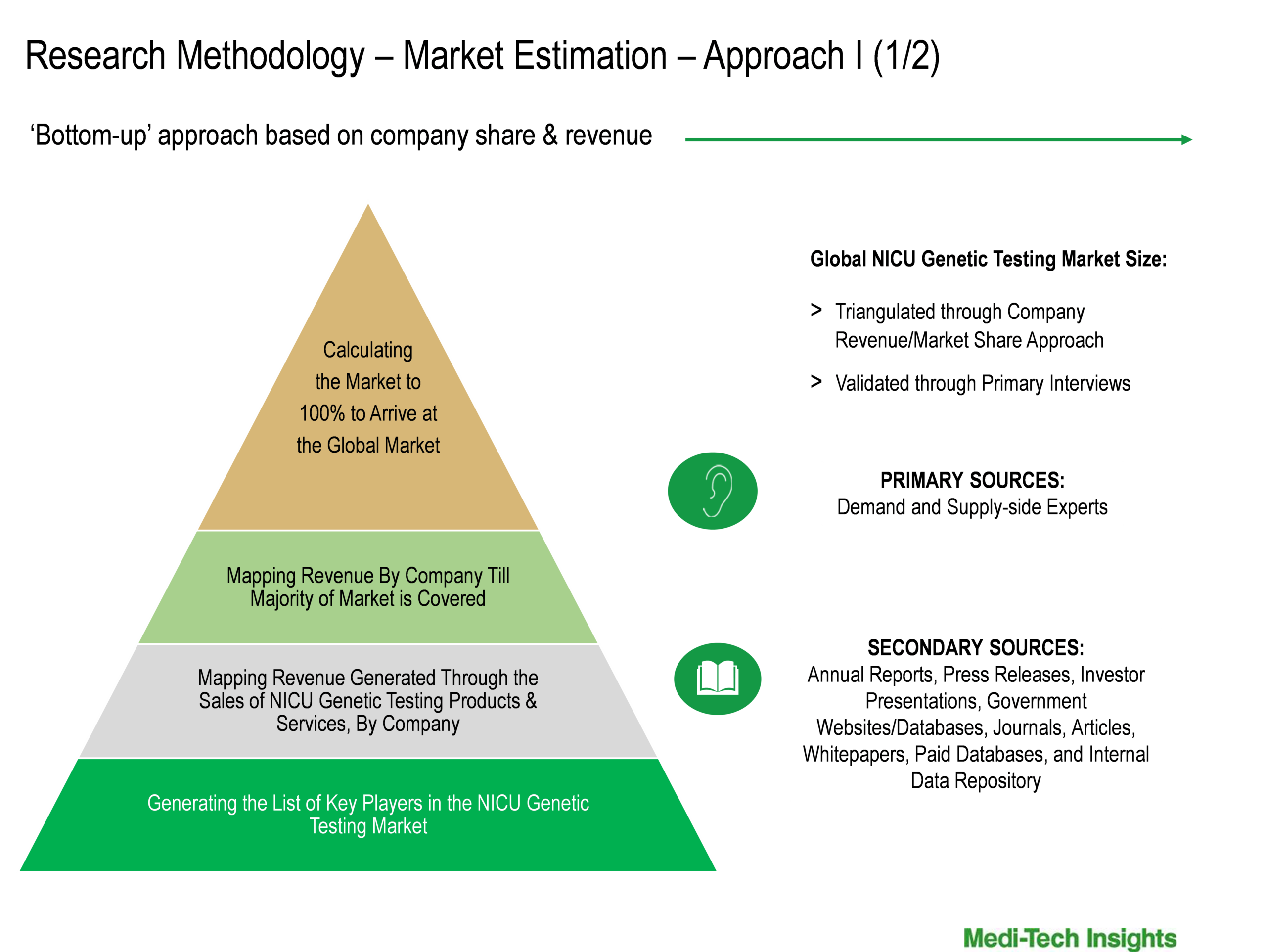
To learn more about this report, download the PDF brochure
Data Triangulation
Research findings derived through secondary sources & internal analysis was validated with Primary Interviews, Internal Knowledge Repository and Company’s Sales Data

Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel