Biosimulation Market (2026-2031) Size, Share, Analysis By Service Type (Software, Molecular Simulation, In-house, Contract Services), Application (Pre-Clinical and Clinical Drug Development, Drug Discovery), End User (Pharmaceutical and Biotechnology Companies, CRO’s), Region and Segment Forecast
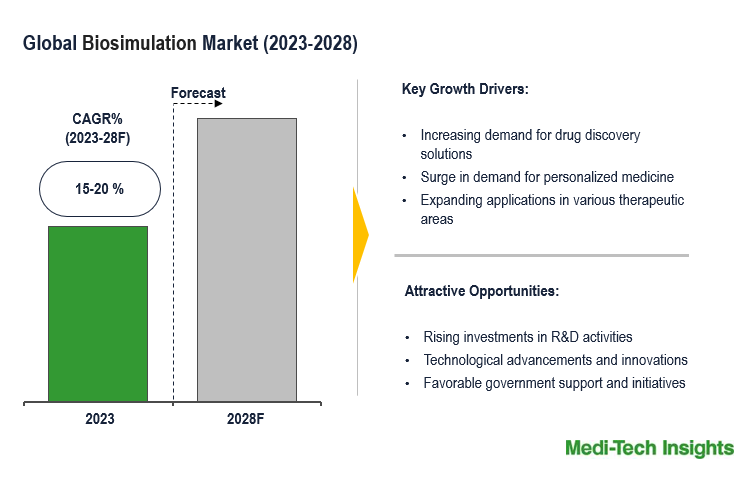
The Global Biosimulation Market will witness a growth rate of 15-20% by 2031. The biosimulation market is driven by factors, including rising demand for drug discovery solutions, the surge in personalized medicine trend, expansion of biosimulation into new therapeutic areas, increasing number of R&D investments, integration of artificial intelligence and advancements in Quantitative Systems Pharmacology (QSP) systems. To learn more about the research report, download a sample report.
Biosimulation, employed in biology and medicine, utilizes computational methods to mimic biological processes. It entails the development of mathematical models replicating the behaviour of biological systems, spanning from cells and tissues to entire organisms. These models are employed to forecast the response of these systems under varying conditions, such as alterations in drug dosage or environmental factors. Biosimulation finds application in drug discovery, personalized medicine, and understanding disease mechanisms, offering insights not easily accessible through traditional experimental methods alone. This approach aids in expediting drug development, cutting costs, and minimizing reliance on traditional animal testing or trials.
Precision Medicine and AI: Revolutionizing Drug Development through Biosimulation
The current landscape of medicine is witnessing a profound shift towards personalized treatment approaches, departing from conventional treatments or methodologies. Precision medicine, fueled by genetic and biomarker data, customizes treatment decisions for individual patients, accounting for genetic variations, environmental factors, and lifestyle choices. This data-driven approach is further augmented by AI-driven biosimulation technologies, revolutionizing drug development processes. Biosimulation, leveraging AI and machine learning, aids in de-risking research and development decisions by offering early, precise, and scalable insights. Notably, AI-enabled virtual simulations are replacing conventional testing methods, expediting drug discovery, addressing ethical concerns, and optimizing outcomes. These advancements not only accelerate drug development by eliminating unnecessary animal trials but also offer cost-effective alternatives and foster regulatory compliance, thus reshaping the future of medicine. For instance,
- In June 2022, Genedata AG announced that Syros Pharmaceuticals had chosen Genedata AG's Genedata Profiler as its platform for data integration and exploration, to accelerate Syros Pharmaceuticals' translational research strategy
- In March 2022, ACD/Labs revealed that Pharmaron Inc. had opted to provide their scientists with ACD/Labs' top-tier NMR processing and interpretation software
To learn more about this report, download the PDF brochure
Biosimulation: Pioneering New Frontiers in Therapeutics and Research
Biosimulation is undergoing a notable expansion, moving beyond conventional therapeutic areas like oncology and cardiovascular diseases to encompass emerging fields such as neurology, infectious diseases, and rare disorders, creating novel avenues for growth. Key trends include the rising adoption of biosimulation in virtual clinical trials, enabling in silico trial conduct that reduces costs and expedites drug development. Moreover, there is a growing inclination towards modeling complex biological systems at various levels, from molecular interactions to organ-level physiology, allowing for more comprehensive simulations and predictions. In silico pharmacology, utilizing computational models to forecast drug responses and toxicity, is gaining momentum, offering early optimization of drug efficacy and safety profiles. Biosimulation is revolutionizing drug development with diverse applications, encompassing pharmacokinetic modeling for drug ADME prediction, pharmacodynamics modeling for understanding molecular interactions and drug efficacy, safety assessment for early identification of potential adverse effects, and biological systems modeling providing insights into disease mechanisms and facilitating the development of innovative therapies. For instance
- In November 2023, Certara, Inc. unveiled the Simcyp™ Biopharmaceutics software which is part of the Simcyp Simulator biosimulation platform, designed to assist biopharmaceutic, formulation, and CMC scientists in formulating complex novel and generic small molecule medicines quickly and cost-effectively.
- In October 2023, Medidata, a company under Dassault Systèmes, revealed the expansion of their multi-year partnership with Catalyst Clinical Research to enhance support for their global oncology brand, Catalyst Oncology
Biosimulation Market: Key Constraints/Challenges
The biosimulation market faces hurdles due to a lack of standardization, complexity in modeling biological systems accurately, and scarcity of experts. Moreover, validating and verifying biosimulation models against real-world data is challenging due to the diverse and dynamic behaviours exhibited by biological systems, posing additional obstacles to comprehensive simulation. These obstacles hinder market growth, emphasizing the need for standardized practices, enhanced expertise, and improved validation techniques.
North America Accounts for the Largest Share of the Global Market
The biosimulation market is regionally segmented, with North America leading due to increasing collaborations among key players to enhance drug discovery capabilities. Additionally, the region's market growth is driven by the utilization of in-silico models for regulatory policy enforcement, ensuring high patient safety and treatment standards. In the Asia Pacific region, significant growth is anticipated, attributed to the rising preference for contract research organizations and increasing healthcare IT expenditure. Moreover, continual enhancements in healthcare infrastructure and heightened research and development activities in developing countries are expected to fuel market growth in the region in the foreseeable future.
Biosimulation Market: Competitive Landscape
Some of the key players operating in the market are Certara, Simulations Plus, Dassault Systèmes, Schrödinger, Inc., ACD/Labs, Chemical Computing Group, Physiomics, Evidera, In silico biosciences, INOSIM Software, Insilico Biotechnology, and Nuventra Pharma among others.
Get a Sample Report for Competitive Landscape Analysis
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as collaborations, and acquisitions to garner market share. For instance,
- In December 2023, Certara completed the acquisition of Applied BioMath, broadening its biosimulation portfolio to enhance capabilities in optimizing dosing for novel therapies. The merger establishes the largest quantitative systems pharmacology (QSP) center of excellence in the life sciences industry, aiming to accelerate and improve the development of medicines
- In September 2023, Genedata revealed a collaboration with the Astellas Institute for Regenerative Medicine (AIRM), a subsidiary of Astellas Pharma Inc., where AIRM licensed Genedata Selector. This partnership aims to ensure the biosafety of cell therapies using Next-Generation Sequencing (NGS)-based assays in a validated environment, thus facilitating regenerative cell therapy product development
The biosimulation market is expected to gain further momentum in the coming years due to the increasing demand for drug discovery, growing demand for personalized medicine, technological advancements, and aggressive organic and inorganic growth strategies followed by the players.
Key Strategic Questions Addressed
- What is the market size & forecast for the Global Biosimulation Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global Biosimulation Market?
- How has COVID-19 impacted the Global Biosimulation Market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the market?
- What is the investment landscape?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the market?
- What are the key strategies adopted by players?
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Market Dynamics
- Global Biosimulation Market - Size & Forecast (2026-2031), By Product and Service Type
- Software
- Molecular Simulation
- In-house
- Contract Services
- Others
- Global Biosimulation Market - Size & Forecast (2026-2031), By Application
- Pre-Clinical and Clinical Drug Development
- Drug Discovery
- Others
- Global Biosimulation Market - Size & Forecast (2026-2031), By End User
- Pharmaceutical and Biotechnology Companies
- CRO’s
- Other End Users
- Global Biosimulation Market - Size & Forecast (2026-2031), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Competitive Positioning of Key Players (2022)
- Offerings Assessment, By Players
- Key Strategies Assessment, By Player (2021-2023)
- New Product Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Other Developments
- Key Players and their Competitive Positioning
- Key Companies Scanned (Indicative List)
- Certara
- Simulations Plus
- Dassault Systemes
- Schrodinger, Inc.
- ACD/Labs
- Chemical Computing Group
- Physiomics
- Evidera
- In Silico Biosciences
- INOSIM Software
- Insilico Biotechnology
- Nuventra Pharma
- Other Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Pharmaceutical & Biotechnology Companies, and Contract Research Organisations among others
Breakdown of Primary Interviews
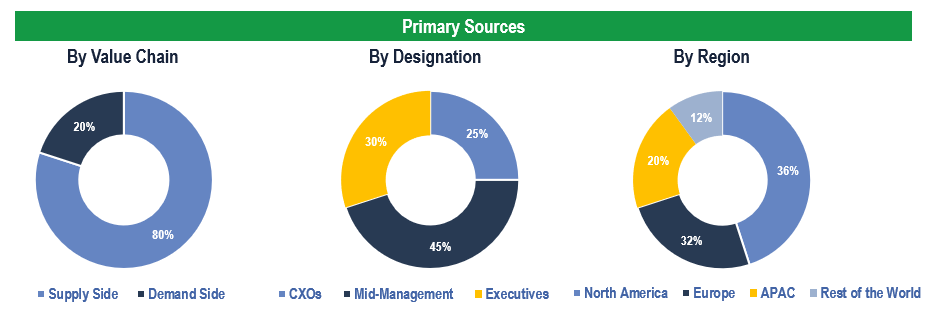
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel