
Global Cancer Biomarkers Market Size, Share & Industry Analysis Report By Type (Breast, Prostate, Colon, Thyroid), By Biomolecule (Genetic, Epigenetic, Metabolic, Proteomic), By Application, Technology, and Region Forecasts, 2026–2031
The Global Cancer Biomarkers Market is set to witness a growth rate of 6-7% by 2031.Growing use of cancer biomarkers in drug discovery and development, favorable funding related to cancer biomarkers, growing research on cancer biomarkers, technological advancements in the development of cancer biomarkers, and an increasing burden of cancer are some of the key factors driving the market growth. To learn more about the research report, download a sample report.
Cancer biomarkers are biological molecules that indicate the presence of cancer in a patient. They are produced either by the cancer cells or by noncancer cells in response to cancer.
Use of Cancer Biomarkers Across Different Stages of Cancer Fuels Its Demand
Cancer biomarkers play a critical role in accurately evaluating and managing cancer in different stages. They are used for cancer risk assessment, screening, differential diagnosis, prognosis determination, treatment response prediction, pharmacokinetics/predicting drug doses, and monitoring treatment response. Clinical use of biomarkers for various types of cancer - breast cancer, prostate cancer, ovarian cancer, pancreatic cancer, colon cancer germ cell cancer, and thyroid cancer, amongst others drives its demand.
Growing Prominence of Cancer Biomarkers in Drug Discovery, Development, and Approval Processes
Biomarkers are increasingly being used at every stage of drug discovery and development. The utilization of biomarkers makes the drug discovery, development, and approval processes more efficient. For the successful treatment of complex chronic diseases (such as cancer), to develop precision medicine and targeted therapies, it is critical to monitor the molecular basis of disease initiation and progression before, during, and after the therapeutic intervention. The need to better understand the factors affecting response to drugs has led to an increased demand for additional efficacy, specificity, and safety testing of drugs during new drug development which in turn has bolstered the demand for cancer biomarkers.
Favorable Funding Environment Set to Drive the Cancer Biomarkers Market Growth
To alleviate the burden of cancer, organizations across the globe are offering funding/grants to discover cancer biomarkers that can treat diseases with unmet needs. For instance,
- Cancer Research UK is offering to fund researchers interested in ‘Biomarker Research’. Through their funding scheme ‘Biomarker Project Awards’, Cancer Research UK is offering 3-year funding of up to £100,000 per year for biomarker assay development, validation, and qualification where there is a clear line of sight to clinical implementation
- Similarly, the National Institutes of Health (NIH) - Cancer Biomarkers Research Group is offering several funding opportunities for cancer biomarkers
- In 2021, Wayne State University researchers received Department of Defense (DoD) funding for the prostate cancer biomarker discovery project. The three-year project received $863,222 as a DoD Idea Development Award.
- In 2018, Fred Hutchinson Cancer Research Center biostatistician Dr. James Dai was recently awarded a $2.1 million grant from the National Cancer Institute for cancer biomarker research
- In 2018, The US National Cancer Institute awarded $6.5 million in funding (over seven years) to Professor Arul Chinnaiyan, from the University of Michigan to identify cancer biomarkers to improve diagnosis and develop new targeted therapies
- In 2018, the National Institutes of Health (NIH) awarded a $6.4 million grant to Albert Einstein College of Medicine and Hackensack University Medical Center for breast cancer biomarker research
“Cancer biomarker testing is revolutionizing cancer treatment for several types of tumors. The moment we identify a key biomarker and figure out what is driving tumor growth, we can develop treatment strategies that particularly target the tumor abnormality and eventually improve patient outcomes.” - Director, Leading Cancer Center, United States
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in Cancer Biomarkers Market
The cancer biomarkers market is an emerging market marked by the presence of both established and new players. Players operating in the market adopt both organic and inorganic growth strategies such as new product/program launches, partnerships, and acquisitions to garner market share. For instance,
- In March 2022, Vela Diagnostics launched new panels to detect cancer biomarkers
- In March 2022, SYNLAB partnered with OncoDNA to provide comprehensive biomarker testing for cancer therapy selection in Germany
- In February 2022, OncoDNA launched the OncoDEEP Kit for comprehensive biomarker testing of solid tumors
- In May 2021, GE Healthcare acquired oncology biomarker maker Zionexa. GE Healthcare will develop and commercialize the Zionexa biomarkers and PET imaging agent, Cerianna
- In April 2021, The Cancer Support Community (CSC) launched a new biomarker testing tool for lung cancer patients. The online tool empowers lung cancer patients and caregivers in the treatment decision-making process
- In April 2021, Amgen launched Biomarker Assist, a program to help more patients with metastatic (stage IV) non-small cell lung cancer (NSCLC) gain access to biomarker testing
Future Outlook: Global Cancer Biomarkers Market
The global cancer biomarkers market is a promising market that is expected to gain further momentum in the coming years due to technological advancements in genomic and proteomic technologies (gene array technology, improved two-dimensional gel electrophoresis, new mass spectrometric techniques) and bioinformatic tools that showcase the potential to meet the growing demand for new biomarkers that are both sensitive and specific.
To ensure precise evaluation and better clinical cancer management, the demand for prognostic and predictive cancer biomarkers is expected to increase. With the growing development of new treatments, there is a growing requirement for biomarkers that can predict outcomes and direct patients/providers on which treatment options are most likely to be effective for a particular patient with a particular tumor.
Competitive Landscape Analysis: Cancer Biomarkers Market
The Cancer Biomarkers Market is marked by the presence of prominent players such as Roche, Thermo Fisher Scientific, QIAGEN, Illumina, Bio-Rad Laboratories, Abbott Laboratories, Sysmex, and others. The foray of players such as R&D Systems, BioVision, Olink, Meso Scale Diagnostics, and others are changing the dynamics of the Cancer Biomarkers market and have made the market more competitive.
Request for Report TOC
Key Strategic Questions Addressed
- What is the market size & forecast of the Cancer Biomarkers Market?
- What are historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Cancer Biomarker Market?
- What are the key trends defining the market?
- What are the major factors impacting the Cancer Biomarkers market?
- What are the opportunities prevailing in the market
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the Cancer Biomarkers market?
- What are the key strategies adopted by players?
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)

Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Academic Institutes, Pharma & Biotechnology Companies, Clinical Research Organizations, Hospitals & Diagnostic Labs.
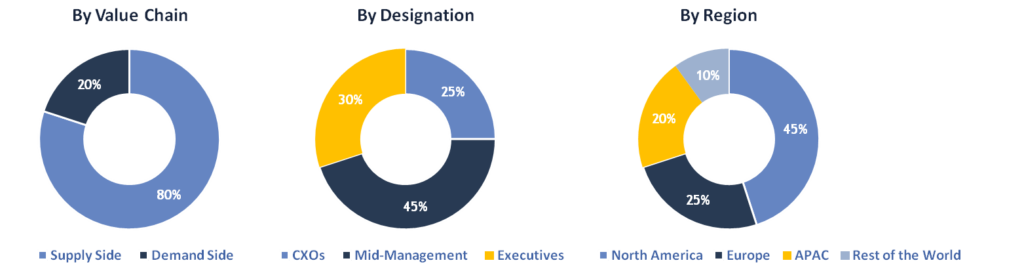
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts
Data Triangulation
Research findings derived through secondary sources & internal analysis was validated with Primary Interviews, Internal Knowledge Repository and Company’s Sales Data
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

