Cardiopulmonary Devices Market Size, Share, Demand, Growth & Applications 2026
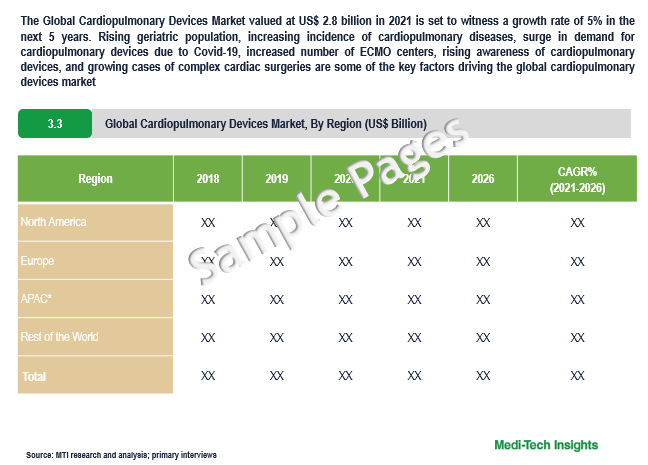
The Global Cardiopulmonary Devices Market valued at $2.8 billion in 2021 is set to witness a growth rate of 5% by 2026. Rising geriatric population, growing number of cardiopulmonary diseases, rise in the demand for cardiopulmonary devices due to Covid-19, growing number of ECMO centers, increasing awareness on cardiopulmonary devices, and growing cases of complex cardiac surgeries are some of the key factors driving the cardiopulmonary devices market growth. However, the dearth of perfusionists in developed regions such as USA is likely to hamper the growth of the market.
Cardiopulmonary diseases include a range of conditions that affect the heart and lungs. Cardiopulmonary devices include heart-lung machines (HLM), percutaneous ventricular assist devices (PVAD), extracorporeal membrane oxygenation (ECMO) & Autotransfusion (ATS). These devices find usage in cardiac surgeries, pulmonary surgeries, congenital defects, cardiopulmonary bypass procedures, cardiogenic shock/respiratory failure, joint replacement surgeries, aneurysm surgeries, transplants, and other indications.
Favourable Impact of Covid-19 on the Cardiopulmonary Devices Market
Throughout 2020 and 2021, the Covid-19 virus spread rapidly and relentlessly across the globe and adversely impacted a multitude of industries worldwide. With lockdowns implemented, social distancing mandated, and widespread apprehension – Covid-19 adversely impacted health services internationally. However, the virus favorably impacted the cardiopulmonary devices market.
During 2020-2021, most of the patients infected with the Covid-19 virus showcased moderate symptoms and recovered quickly. However, some patients developed severe respiratory failure and required intensive care unit (ICU) admission and mechanical ventilation. To treat and save the lives of these severe Covid-19 patients, ECMO emerged as a promising treatment option. The ECMO machine supports both the heart and lungs and stabilizes patients to allow their bodies more time to fight the virus.
Growing awareness about ECMO, HLM & other cardiopulmonary devices and the implementation of new ECMO centers during the COVID-19 pandemic further boosted the global cardiopulmonary devices market.
Favorable Regulatory Policies in Key Markets Boosted the Cardiopulmonary Devices Market
To fight the Covid-19 pandemic and to offer cardiopulmonary and advanced circulatory support products and therapies that may benefit Covid-19 virus-infected patients, in April 2020 the FDA permitted manufacturers of cardiopulmonary bypass devices to modify the product indications for use to include ECMO therapy for greater than six hours, without prior submission of a premarket notification to FDA. The favorable regulatory policy change resulted in increased sales of cardiopulmonary and advanced circulatory support products.
“ECMO has emerged as a game-changer for Covid-19 patients. It has proven to be a life saver. More than 200 patients at our hospital who have been on ECMO longer than 24 hours have recovered.” - Director, Mechanical Circulatory Support & ECMO, Leading Hospital, United States
Technological Advancements Drives the Cardiopulmonary Devices Market
The global cardiopulmonary devices market is a technology-driven market and is marked by constant product enhancements/innovations. For instance,
- In April, 2022, LivaNova initiates the targeted commercial launch of the Essenz™ Patient Monitor for cardiopulmonary bypass procedures. The transformative monitoring system uses a patient-tailored approach to improve clinical efficiency and quality of patient care during cardiopulmonary bypass (CPB) procedures.
- In January 2021, Eurosets announced the full commercial release (out of USA) of its novel ECMOlife system for ExtraCorporeal Life Support (ECLS). ECMOlife expands Eurosets’ portfolio to provide temporary support to patients in different therapeutic areas, including ExtraCorporeal Membrane Oxygenation (ECMO) and Mechanical Circulatory Support (MCS).
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold
The cardiopulmonary devices market is marked by the presence of both established and new players. Players operating in the market adopt both organic and inorganic growth strategies such as acquisitions, and new product launches to garner market share. For instance,
- In February 2022, Getinge acquired 100% of the equity interest in Talis Clinical LLC, a US-based leading innovator of High Acuity cloud-based software solutions. Talis Clinical’s offer is designed to support and document care throughout the general and cardiac perioperative care process, but also through labor and delivery episodes and critical care support including ECMO Therapy.
- In October 2021, MicroPort acquired 100% of the equity held by existing shareholders of Hemovent GmbH. Hemovent specializes in the development of ground-breaking Extracorporeal Life Support (ECLS) systems.
- In April 2020, Abiomed, manufacturer of the Impella heart pump, acquired Breethe, developer of a novel extracorporeal membrane oxygenation (ECMO) system. The acquisition complements and expands Abiomed’s product portfolio to more comprehensively serve the needs of patients whose lungs can no longer provide sufficient oxygenation, including patients suffering from cardiogenic shock or respiratory failure such as due to ARDS, H1N1, SARS, or COVID-19.
The cardiopulmonary devices market is expected to continue to grow in the coming years due to growing cases of cardiopulmonary cases, technological advancements in cardiopulmonary devices, rising number of orthopedic & transplant procedures, and rising aging populations.
Competitive Landscape Analysis: Cardiopulmonary Devices Market
The global cardiopulmonary devices market is marked by the presence of leading players such as LivaNova, Getinge, Terumo, Medtronic, Abbott, Abiomed, Haemonetics, Fresenius, among others.
Key Strategic Questions Addressed
- What is the market size & forecast of the Cardiopulmonary Devices Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Cardiopulmonary Devices Market?
- What are the key trends defining the cardiopulmonary devices market?
- What are the major factors impacting the market?
- What are the opportunities prevailing in the cardiopulmonary devices market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the cardiopulmonary devices market?
- What are the key strategies adopted by players operating in cardiopulmonary devices market?
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)

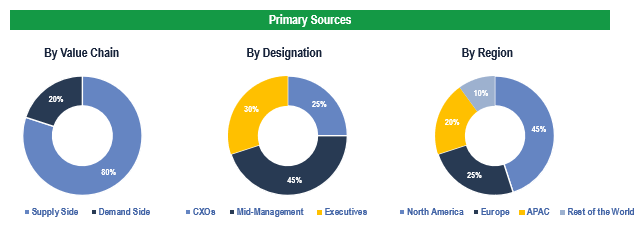
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Ambulatory Surgery Centers, among others.
Breakdown of Primary Interviews
Market Size Estimation
The market size was derived based on extensive secondary research further validated through expert interviews.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with primary interviews and an internal knowledge repository.