Digital Biomarkers Market Size Report Segmented by Product (Wearables, Mobile Applications, Sensors, Software), Application (Diagnosis, Monitoring), Therapeutic Area (CVDs, Psychiatric & Behavioural Health) Forecast to 2030
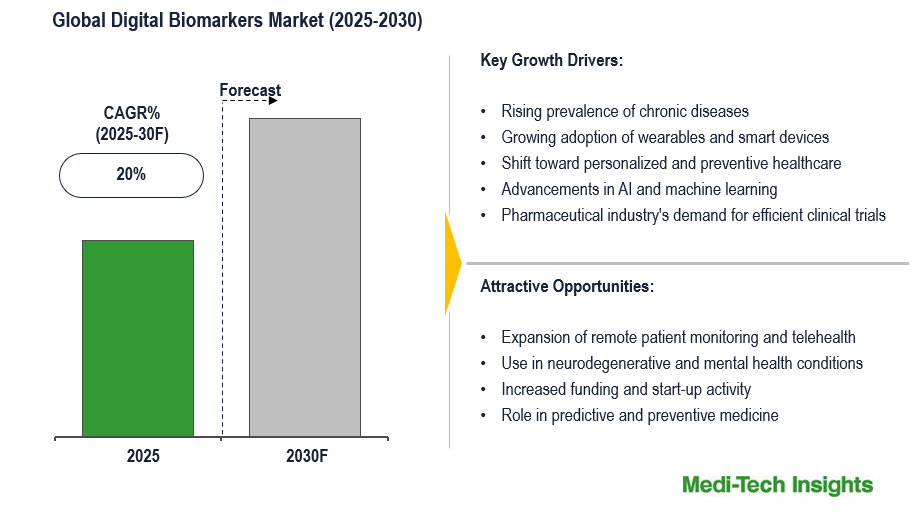
The global digital biomarkers market is set to witness a growth rate of 20% in the next 5 years. Rising prevalence of chronic diseases, growing adoption of wearables and smart devices, shift toward personalized and preventive healthcare, advancements in AI and machine learning, and pharmaceutical industry’s demand for efficient clinical trials, are some of the key factors driving the digital biomarkers market. To learn more about the research report, download a sample report.
Report Overview
Digital biomarkers are objective, quantifiable physiological and behavioral data collected and measured through digital devices such as smartphones, wearables, and sensors. These biomarkers are used to monitor, predict, or diagnose health conditions in real time, providing continuous insights into a patient’s status outside traditional clinical settings. They are particularly useful in chronic disease management, mental health, and neurodegenerative disorders among others. Unlike traditional biomarkers that often require lab-based tests, digital biomarkers enable passive, non-invasive data collection. Advances in artificial intelligence and data analytics further enhance their utility, making them valuable tools for personalized medicine, remote monitoring, and efficient clinical trial design.
To learn more about this report, download the PDF brochure
Growing adoption of wearables and smart devices to propel market demand
The growing adoption of wearables and smart devices is a major driver of the digital biomarkers market. Devices like smartwatches, fitness trackers, and mobile health apps enable continuous, real-time monitoring of physiological and behavioral data such as heart rate, sleep patterns, activity levels, and more. This constant data flow allows for early detection of health anomalies and supports proactive, personalized healthcare. As consumers become more health-conscious and comfortable with digital technologies, the widespread use of these devices fuels demand for digital biomarkers. Additionally, their integration with cloud platforms and AI analytics enhances clinical decision-making and supports remote patient monitoring.
Digital biomarkers powering the shift to decentralized clinical trials
The growth of remote and decentralized clinical trials is a key trend driving the digital biomarkers market. These trials leverage digital biomarkers to collect continuous, real-time patient data outside traditional clinical settings, reducing the need for in-person visits. This approach enhances patient recruitment, retention, and compliance while lowering operational costs. Wearables, mobile apps, and sensors enable seamless remote monitoring, making trials more inclusive and geographically flexible. As regulatory bodies increasingly recognize digital endpoints, the adoption of decentralized trial models is accelerating, positioning digital biomarkers as essential tools in transforming clinical research into a more patient-centric process.

To learn more about this report, download the PDF brochure
Competitive Landscape Analysis
The global digital biomarkers market is marked by the presence of established and emerging market players such as BioSensics, ActiGraph, LLC; AliveCor, Inc.; Koneksa Health; Altoida; Amgen Inc.; Biogen; Empatica Inc.; IXICO plc; Neurotrack Technologies, Inc.; among others. Some of the key strategies adopted by market players include new product development, strategic partnerships and collaborations, and geographic expansion.
Report Scope
| Report Metric | Details |
| Base Year Considered | 2024 |
| Historical Data | 2023 - 2024 |
| Forecast Period | 2025 - 2030 |
| Growth Rate | 20% |
| Market Drivers |
|
| Attractive Opportunities |
|
| Segment Scope | Product, Application, Therapeutic Area, End User |
| Regional Scope |
|
| Key Companies Mapped | BioSensics, ActiGraph, LLC; AliveCor, Inc.; Koneksa Health; Altoida; Amgen Inc.; Biogen; Empatica Inc.; IXICO plc; Neurotrack Technologies, Inc., among others |
| Report Highlights | Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Global Digital Biomarkers Market Segmentation
This report by Medi-Tech Insights provides the size of the global digital biomarkers market at the regional- and country-level from 2023 to 2030. The report further segments the market based on product, application, therapeutic area, end user.
Market Size & Forecast (2023-2030), By Product, USD Million
- Wearable Devices
- Mobile Applications
- Sensors
- Software & Platforms
Market Size & Forecast (2023-2030), By Application, USD Million
- Disease Diagnosis
- Monitoring & Management
- Drug Development & Clinical Trials
- Predictive Risk Analysis
- Behavioural & Cognitive Assessment
- Others
Market Size & Forecast (2023-2030), By Therapeutic Area, USD Million
- Neurological Disorders
- Cardiovascular Diseases
- Diabetes & Metabolic Disorders
- Respiratory Diseases
- Psychiatric & Behavioural Health
- Sleep Disorders
- Others
Market Size & Forecast (2023-2030), By End User, USD Million
- Pharmaceutical & Biotech Companies
- Healthcare Providers
- Healthcare Payers
- Others
Market Size & Forecast (2023-2030), By Region, USD Million
- North America
- US
- Canada
- Europe
- UK
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Middle East & Africa
Key Strategic Questions Addressed
- What is the market size & forecast of the digital biomarkers market?
- What are historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the digital biomarkers market?
- What are the key trends defining the market?
- What are the major factors impacting the market?
- What are the opportunities prevailing in the market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market?
- What are the key strategies adopted by players?
- Introduction
- Introduction
- Market Scope
- Market Definition
- Segments Covered
- Regional Segmentation
- Research Timeframe
- Currency Considered
- Study Limitations
- Stakeholders
- List of Abbreviations
- Key Conferences and Events (2025-2026)
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Bottom-Up Approach
- Top-Down Approach
- Market Forecasting
- Executive Summary
- Digital Biomarkers Market Snapshot (2025-2030)
- Segment Overview
- Regional Snapshot
- Competitive Insights
- Market Overview
- Market Dynamics
- Drivers
- Rising prevalence of chronic diseases
- Growing adoption of wearables and smart devices
- Shift toward personalized and preventive healthcare
- Advancements in AI and machine learning
- Pharmaceutical industry's demand for efficient clinical trials
- Restraints
- Data privacy and security concerns
- Lack of standardization and validation protocols
- Limited reimbursement and coverage policies
- Integration challenges with existing healthcare systems
- Opportunities
- Expansion of remote patient monitoring and telehealth
- Use in neurodegenerative and mental health conditions
- Increased funding and start-up activity
- Role in predictive and preventive medicine
- Key Market Trends
- Growth of remote and decentralized clinical trials
- Collaborations between pharma and tech companies
- Focus on real-world evidence (RWE)
- Unmet Market Needs
- Industry Speaks
- Drivers
- Market Dynamics
- Global Digital Biomarkers Market Size & Forecast (2023-2030), By Product, USD Million
- Introduction
- Wearable Devices
- Mobile Applications
- Sensors
- Software & Platforms
- Global Digital Biomarkers Market Size & Forecast (2023-2030), By Application, USD Million
- Introduction
- Disease Diagnosis
- Monitoring & Management
- Drug Development & Clinical Trials
- Predictive Risk Analysis
- Behavioural & Cognitive Assessment
- Others
- Global Digital Biomarkers Market Size & Forecast (2023-2030), By Therapeutic Area, USD Million
- Introduction
- Neurological Disorders
- Cardiovascular Diseases
- Diabetes & Metabolic Disorders
- Respiratory Diseases
- Psychiatric & Behavioural Health
- Sleep Disorders
- Others
- Global Digital Biomarkers Market Size & Forecast (2023-2030), By End User, USD Million
- Introduction
- Pharmaceutical & Biotech Companies
- Healthcare Providers
- Healthcare Payers
- Others
- Global Digital Biomarkers Market Size & Forecast (2023-2030), By Region, USD Million
- Introduction
- North America Digital Biomarkers Market Size & Forecast (2023-2030), By Country, USD Million
- US
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Canada
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- US
- Europe Digital Biomarkers Market Size & Forecast (2023-2030), By Country, USD Million
- UK
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Germany
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- France
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Italy
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Spain
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Rest of Europe
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- UK
- Asia Pacific (APAC) Digital Biomarkers Market Size & Forecast (2023-2030), By Country, USD Million
- China
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Japan
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- India
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Rest of Asia Pacific
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- China
- Latin America (LATAM) Digital Biomarkers Market Size & Forecast (2023-2030), USD Million
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Middle East & Africa (MEA) Digital Biomarkers Market Size & Forecast (2023-2030), USD Million
- Market Size & Forecast, By Product (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By Therapeutic Area (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Key Player Comparison
- Segment-wise Player Mapping
- Market Share Analysis (2024)
- Company Categorization Matrix
- Dominants/Leaders
- New Entrants
- Emerging Players
- Innovative Players
- Key Strategies Assessment, By Player (2022-2025)
- New Product Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Company Profiles*
(Business Overview, Financial Performance**, Products Offered, Recent Developments)
- BioSensics
- ActiGraph, LLC.
- AliveCor, Inc.
- Koneksa Health
- Altoida
- Amgen Inc.
- Biogen
- Empatica Inc.
- IXICO plc
- Neurotrack Technologies, Inc.
- Other Prominent Players
Note: *Indicative list
**For listed companies
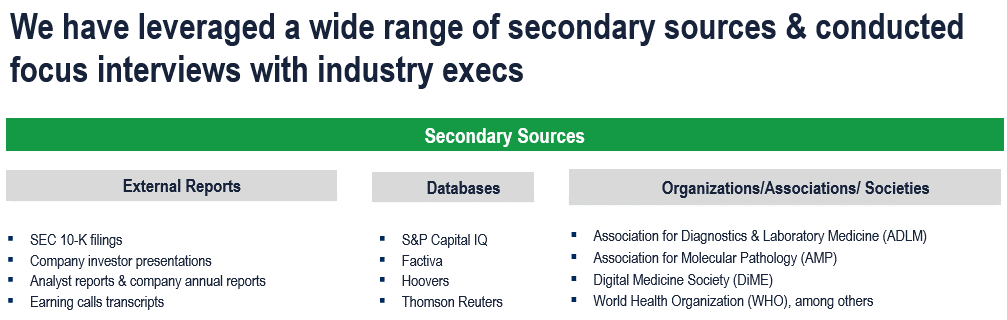
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
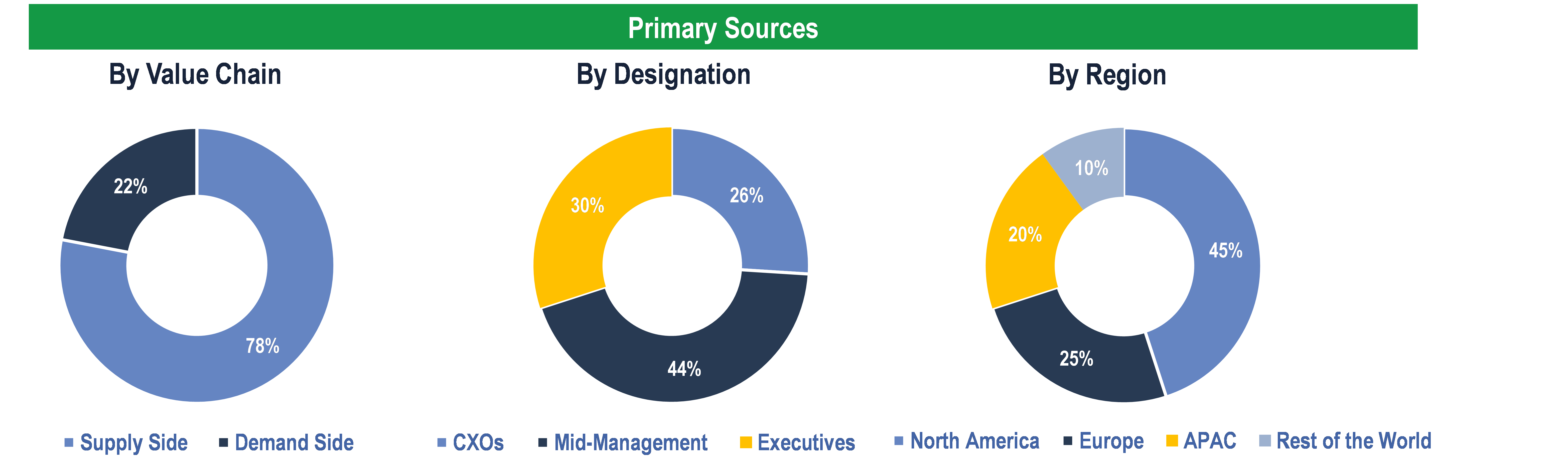
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders from Pharmaceutical & Biotech Companies; Healthcare Providers; Healthcare Payers; and Others
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down & Bottom-Up Approaches’ were used to derive market size estimates and forecasts
Data Triangulation
Research findings derived through secondary sources & internal analysis was validated with Primary Interviews, Internal Knowledge Repository and Company’s Sales Data
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel