e-Prescribing Market – Global Industry Analysis, Size, Growth, Key Drivers and Challenges for Forecast 2024 to 2029
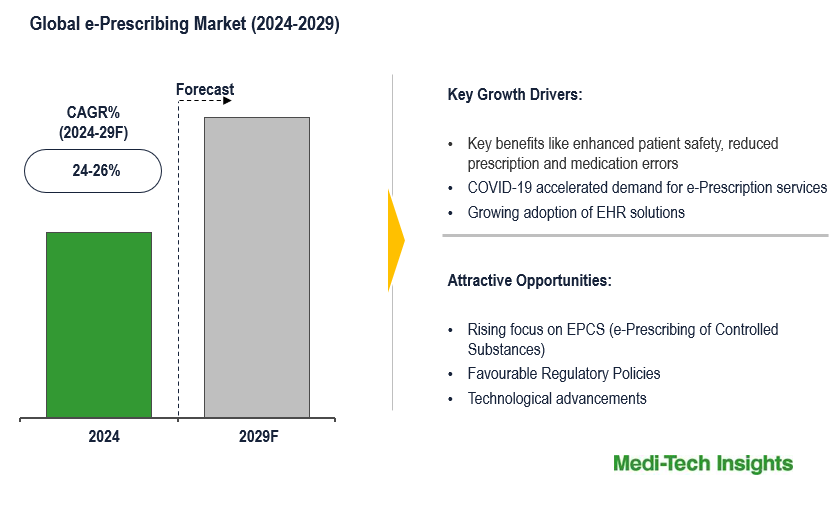
The Global e-prescribing Market is set to witness an exponential growth rate of 24-26% by 2029. Key benefits of e-prescribing (such as enhanced patient safety, reduced prescription and medication errors, instant clinical alerts, prescription fulfillment tracking, etc.), Covid-19, favourable regulatory environment in selected countries, growing use of telemedicine, the emerging focus on reducing the abuse of controlled substances, adoption of EHR solutions, government initiatives and incentive programs are some of the key factors driving the market growth. However, limited technology awareness among end-users is likely to hamper the e-prescribing market growth. To learn more about the research report, download a sample report.
e-Prescribing is the prescriber's ability to electronically send an accurate, error-free, and understandable prescription directly to a pharmacy from the point of care.
Covid-19 Triggers Increase in e-Prescribing Market
Throughout 2020 and 2021, the COVID-19 virus spread rapidly and relentlessly across the globe and adversely impacted a multitude of industries worldwide. With lockdowns implemented, social distancing mandated, and widespread apprehension – COVID-19 adversely impacted health services internationally and healthcare providers were compelled to change their methods of service delivery. In the wake of the COVID-19 pandemic, e-Prescribing emerged as an important tool in creating a safe environment to avoid the spread of COVID-19, minimizing in-person consultations and paper prescription handling.
According to Surescripts (a US-based information technology company that supports e-prescriptions), Covid-19 sparked a 72.5% increase in the number of electronic prescriptions written through telehealth during the first month of Covid-19.
Favorable Regulatory Changes Across Countries Provide Impetus to the e-Prescribing Market
Healthcare delivery including pharmacy services is highly regulated worldwide, to protect public health and welfare. However, the COVID-19 pandemic has triggered the need to amend the e-prescription legislation so that people can access a range of healthcare services in a timely and effective manner. For instance,
- To curb the rise in drug-related deaths in the US, the federal government and several states including California, Indiana, Maryland, Michigan, Nebraska, New Hampshire, Utah, and Washington have mandated the e-prescribing of controlled medications in the US. The mandate requires clinical prescribers to write controlled medications electronically
- In Ireland, The Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations 2020 allowed the electronic transfer of prescriptions to a pharmacy via an approved electronic system: the HSE's Healthmail system. The legislation removed the requirement for a physical or hard copy of a prescription to be presented in pharmacies to collect prescriptions that are sent via the HSE’s secure Healthmail service. Additionally, the maximum period of validity of a prescription was increased from 6 months to 9 months as of the date specified on the prescription
- Similarly, the Covid-19 pandemic accelerated work related to e-prescriptions and e-prescriptions are now available across Australia. The Australian Government changed its legislation to make electronic prescribing of Pharmaceutical Benefits Scheme (PBS) medicines legal. States and territories also made changes to their legal frameworks to allow for electronic prescriptions in their jurisdiction
- In August 2023, DrFirst Healthcare Innovations announced a collaboration with MEDITECH to enable electronic prescribing for hospitals using the Expanse EHR in Canada. This integration aligns with MEDITECH’s plans to connect with Canada Health Infoway’s national e-prescribing service, PrescribeIT.
To learn more about this report, download the PDF brochure
e-Prescription Benefits Fuels It’s Demand
Increasing usage of technology across every aspect of healthcare has enabled the demand for e-prescription globally.
Some of the key benefits of e-prescribing that are driving its demand are:
- Reduced Pharmacist Error: As compared to hand-written prescriptions, e-prescriptions are explicit and clear in terms of prescribing treatment and dosage. This leads to reduced prescription and medication errors as it requires less interpretation from pharmacists
- Instant Clinical Notifications: e-Prescribing systems provide notifications to prescribers about allergies, potential drug interactions, duplicate therapies, pregnancy and other issues that would be contraindicated for a patient when taking a particular medication
- Prescription Fulfillment Tracking: e-prescription systems help doctors/clinicians track whether or not patients are adhering to prescribed medication/treatment
- Reconcile Medication History Instantly: Using e-prescription systems, prescribers can view patient’s medication history instantly which in turn saves medication lists reconciling time
- Improved Prescription Efficiency: Using advanced e-prescription systems, clinicians can automatically refill several prescriptions at the same time, which allows them more time to review. This is likely to increase both efficiency and reduce the prescription error rate
- Reduced Lost Prescriptions: Hand-written paper prescriptions are small and easy to misplace which increases burden on medical offices as they need to rewrite or call the prescriptions into the pharmacy. e-Prescription can be sent directly to the pharmacy, so patients do not have to keep track of hand-written paper prescriptions.
“Electronic Prescribing for Controlled Substances (EPCS) mandate in the United States allows clinical prescribers to write prescriptions electronically for controlled substances. More than half of the US falls under EPCS mandates. It is a vital tool to reduce unwarranted deaths caused due to drug overdose. Growing EPCS mandate in US states is likely to drive the e-prescribing market.” - Senior Director, Leading Electronic Prescribing Service Provider, United States
Product Segment Outlook
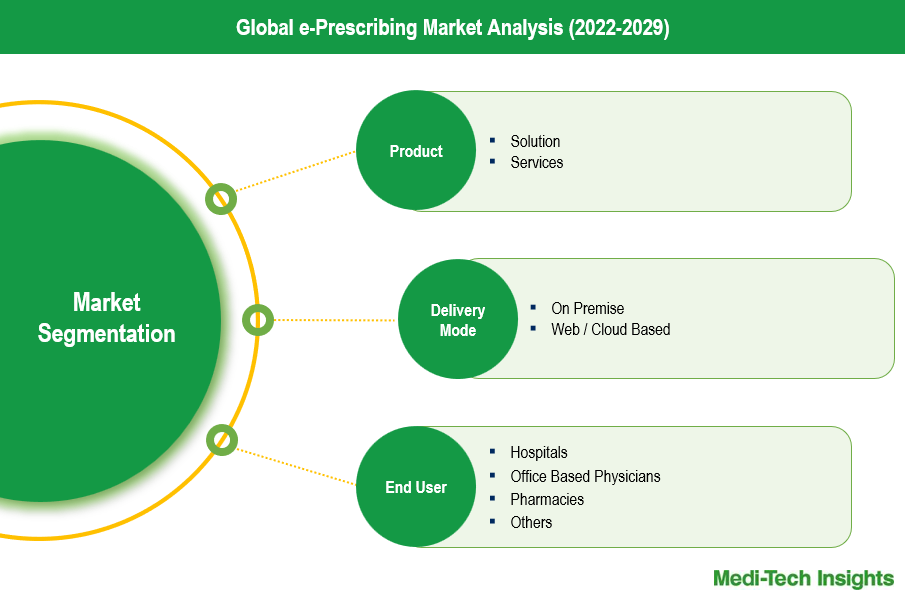
The e-prescribing market is segmented into services and solutions, each playing a crucial role in the market’s expansion and holding distinct market shares. Solutions encompass software platforms and applications that facilitate the electronic transmission of prescriptions from healthcare providers to pharmacies. These solutions are integral to the e-prescribing ecosystem, offering clinical decision support, medication history tracking, and prescription fulfillment monitoring functionalities. The solutions segment commands a significant market share due to the increasing adoption of electronic health records (EHR) and the growing need for seamless, error-free prescription processes. Services on the other hand, include implementation, maintenance, training, and support services necessary for the effective deployment and operation of e-prescribing systems. This segment is vital for ensuring the optimal performance and compliance of e-prescribing platforms, thereby driving their usability and reliability. Although the services segment holds a smaller market share compared to solutions, it is essential for the sustained adoption and effectiveness of e-prescribing technologies, contributing to the overall growth of the market.
To learn more about this report, download the PDF brochure
End-User Segment Outlook
The e-prescribing market is categorized by end users into hospitals, office-based physicians, and pharmacies, each with distinct market shares and roles in the ecosystem. Hospitals represent a substantial segment due to the high volume of prescriptions generated and the need for integrated healthcare solutions that enhance patient safety and streamline operations. The adoption of e-prescribing in hospitals is driven by the integration of electronic health records (EHR) systems and the need for efficient medication management. Office-based physicians form another significant segment, as they frequently issue prescriptions during outpatient visits. The convenience and efficiency offered by e-prescribing solutions encourage widespread adoption among these healthcare providers, contributing notably to market growth. Pharmacies also play a crucial role, utilizing e-prescribing systems to receive and process electronic prescriptions seamlessly. This reduces prescription errors and enhances operational efficiency. While hospitals and office-based physicians dominate the market share due to their direct interaction with patients and higher prescription volumes, pharmacies are essential for the completion of the e-prescribing process, ensuring the accurate dispensing of medications. Together, these end-user segments drive the comprehensive adoption and expansion of the e-prescribing market.
Competitive Landscape Analysis: e-Prescribing Market
The global e-Prescribing market is marked by the presence of players such as Allscripts, NextGen Healthcare, AdvancedMD, MDToolbox, Practice Fusion, DrFirst, RXNT, Chetu, PrognoCIS, NewCrop, among others.
Get a sample report for competitive landscape analysis
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as collaborations, acquisitions, and new product launches to garner market share. For instance,
- In April 2024, VieCure, a cancer care company, announced the integration of e-prescribing and medication management solutions from health technology pioneer DrFirst into its clinical decision support system
- In November 2023, Thoma Bravo announced the completion of its $1.8 billion acquisition of NextGen Healthcare, Inc., a provider of innovative, cloud-based healthcare technology solutions
- In October 2023, FDB and Veradigm announced that Veradigm, the first major electronic health record (EHR) network to join the FDB Vela™ ePrescribing network, will integrate FDB Vela into its solutions, including EHRs and the Veradigm ePrescribe Enterprise solution, thereby expanding the Veradigm Network's community of companies providing advanced insights, technology, and data-driven solutions
- In March 2022, FDB (First Databank, Inc.), the leading provider of drug and medical device databases, launched FDB Vela, a new, cloud-native electronic prescribing (ePrescribing) network that enables the seamless flow of critical medication prescription information, benefits verification, and clinical decision support between prescribers, payers, pharmacies, and other constituents
- In October 2021, Veradigm (a business unit of Allscripts Healthcare Solutions), and CareMetx, LLC announced a strategic partnership to integrate CareMetx’s solutions and services into the Veradigm AccelRx® speciality medication platform, aiming to simplify and accelerate the specialty medication approval process and optimize end-to-end patient care
The e-Prescribing market is a booming market that is expected to gain further momentum in the coming years due to the benefits of e-prescribing, COVID-19 after-effects, favorable regulatory changes across countries, accelerated use of telemedicine, and growing focus on reducing the abuse of controlled substances.
|
Report Scope |
Details |
|
Base Year Considered |
2023 |
|
Historical Data |
2022 - 2023 |
|
Forecast Period |
2024 - 2029 |
|
CAGR (2024-2029) |
24-26% |
|
Segment Scope |
Product, Delivery Mode, and End User |
|
Regional Scope |
|
|
Key Companies Mapped |
Allscripts, NextGen Healthcare, AdvancedMD, MDToolbox, Practice Fusion, DrFirst, RXNT, Chetu, PrognoCIS, and NewCrop among others |
|
Report Highlights |
Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Key Strategic Questions Addressed
-
What is the market size & forecast for the Global e-Prescribing Market?
-
What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global e-Prescribing Market?
-
How has COVID-19 impacted the Global e-Prescribing Market?
-
What are the major growth drivers, restraints/challenges impacting the market?
-
What are the opportunities prevailing in the market?
-
What is the investment landscape?
-
Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
-
Who are the major players operating in the market? What is the competitive positioning of key players?
-
Who are the new players entering the market?
-
What are the key strategies adopted by players?
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
-
- Market Dynamics
- Drivers
- Restraints
- Key Market Trends
- Industry Speaks
- Market Dynamics
- Key Revenue Pockets
- Global e-Prescribing Market - Size & Forecast (2021-2028), By Product
- Solutions
- Services
- Global e-Prescribing Market - Size & Forecast (2021-2028), By Delivery Mode
- On-Premise
- Web / Cloud Based
- Global e-Prescribing Market - Size & Forecast (2021-2028), By End User
- Hospitals
- Office-Based Physicians
- Pharmacies
- Others
- Global e-Prescribing Market - Size & Forecast (2021-2028), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Competitive Positioning of Key Players (2022)
- Offerings Assessment, By Player
- Key Strategies Assessment, By Player (2021-2023)
- New Product & Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Key Companies Scanned (Indicative List)
- Allscripts
- NextGen Healthcare
- AdvancedMD
- MDToolbox
- Practice Fusion
- DrFirst
- RXNT
- Chetu
- PrognoCIS
- NewCrop
- Other Prominent Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Office-Based Physicians, Pharmacies and Other End Users
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel