
Endoscope Reprocessing Market Share & Growth Analysis, by Product (AER, HLD & Test Strip, Detergent & Wipe, Endoscope Drying, Storage, & Transport System, Tracking Solution), Endoscope Type (Flexible, Rigid), End User (Hospital, ASC, Specialty Clinic) and Regional Forecast to 2031
The Endoscope Reprocessing Market is expected to witness a growth rate of 10% by 2031.Rising number of gastrointestinal (GI), respiratory, ear, urinary tract, and female reproductive tract procedures, increasing preference for minimally invasive surgeries & growing usage of endoscopy for the diagnosis & treatment of several disorders, high risk of endoscope-related infections due to contaminated endoscopes, technological advancements in endoscope reprocessing products, growing hospital investments in endoscopy facilities, roll-out & implementation of reprocessing guidelines for endoscopy units by healthcare authorities are some of the key factors driving the growth of the global endoscope reprocessing market.
However, lack of trained professionals and rising preference for single-use endoscopes are some of the factors which may impact the market’s growth.
Endoscopy is a medical procedure that allows a doctor to inspect and observe the inside of the body without performing major surgery. It is used worldwide for the screening, diagnosis, and treatment of gastrointestinal (GI), respiratory, ear, urinary tract, and female reproductive tract disorders. In an endoscopic examination, the external surface and internal channels of endoscopes are exposed to body fluids and contaminants. Hence, reprocessing endoscopes is imperative for infection prevention. Common steps involved in the reprocessing of endoscopes are precleaning, cleaning, rinsing, disinfection, rinsing, drying, and storage.
The Resurgence of Endoscope Reprocessing Market Post Covid-19 Pandemic
Throughout 2020 and 2021, the Covid-19 virus spread rapidly and relentlessly across the globe and adversely impacted a multitude of industries worldwide. With lockdowns implemented, social distancing mandated, and widespread apprehension – Covid-19 adversely impacted health services internationally and the endoscope reprocessing market was no different.
Specialties such as gastroenterology were directly affected by Covid-19. The pandemic adversely impacted the workflow and safety of endoscopists, ancillary staff, and patients. Shortages of personal protective equipment (PPE), curtailed patient volume, dearth of testing kits, workforce furloughs, and lockdowns forced units to drastically reduce the volume of the procedures, postpone and even cancel GI procedures in medium- to low-risk cases. The dip in the number of endoscopy procedures resulted in a decline in endoscope reprocessing revenue.
However, since 2021, the endoscope reprocessing market is on the recovery path. The market has been driven by increased sales of gastroscopes and colonoscopes, the launch of innovative products, and a rising number of GI, respiratory, ear, urinary tract, and female reproductive tract procedures.
“Due to growing penetration of endoscopes across hospitals globally, the demand for scope reprocessing is rising as it plays a critical role in the fight against health care acquired infections (HCAI) and contributes to a more sustainable health care.” -Senior Director, Leading Automated Endoscope Reprocessor (AER) Manufacturer, United States
Technological Advancements Drive the Endoscope Reprocessing Market
The endoscope reprocessing market is a technology-driven market and is marked by constant product enhancements/innovations. For instance,
- In June, 2022, Getinge launched an updated version of the ED-Flow automated endoscope reprocessor. The new features offer a higher level of digital connectivity and data management which in turn is expected to result in improved uptime and increased productivity
- In September 2021, Steelco launched EW 1 S MAXI, an automated endoscope reprocessor (AER) that incorporates all the latest technologies to increase safety, efficiency, and ease of use in endoscope reprocessing.
United States Holds Major Share in the Endoscope Reprocessing Market
The United States currently holds a major share in the global endoscope reprocessing market. The large share of the North American market can be attributed to an increasing number of endoscopic examinations, high prevalence of hospital-acquired infections, rising aging population, favorable reimbursement climate for endoscopy procedures, growth in minimally invasive endoscopic therapy procedures, rising concerns over cleaning, disinfection & sterilization, and higher quality expectations.
Organic and Inorganic Growth Strategies Adopted by Industry Players to Establish Their Foothold in Endoscope Reprocessing Market
The global endoscope reprocessing market is marked by the presence of both established and new players. Players operating in the market adopt both organic and inorganic growth strategies such as new product launches, and partnerships to garner market share. For instance,
- In January 2021, STERIS entered into a definitive agreement to acquire Cantel. The acquisition of Cantel complements STERIS’s product and service portfolio focused on infection prevention.
- In December 2020, Cantel Medical and Censis Technologies entered into partnership to develop differentiated endoscope reprocessing software solution. As per the terms of the agreement, Cantel's leading infection prevention endoscope reprocessing workflow portfolio will be combined with the surgical asset management and instrument tracking solutions from Censis to provide superior infection prevention and practice optimization.
The endoscope reprocessing market is expected to continue to grow in the coming years due to growing cases of gastrointestinal (GI), respiratory, ear, urinary tract, and female reproductive tract procedures, technological advancements in endoscope reprocessing products, and growing demand for infection prevention during endoscopic examinations.
Competitive Landscape Analysis: Endoscope Reprocessing Market
The global endoscope reprocessing market is marked by the presence of players such as STERIS/Cantel Medical, Wassenburg Medical, Getinge, Advanced Sterilization Products (ASP), Olympus, CONMED, Ecolab, among others.
Request for TOC
Key Strategic Questions Addressed
- What is the market size & forecast of the Endoscope Reprocessing Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Endoscope Reprocessing Market?
- What are the key trends defining the endoscope reprocessing market?
- What are the major factors impacting the endoscope reprocessing market?
- What are the opportunities prevailing in the endoscope reprocessing market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the endoscope reprocessing market?
- What are the key strategies adopted by players operating in endoscope reprocessing market?
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
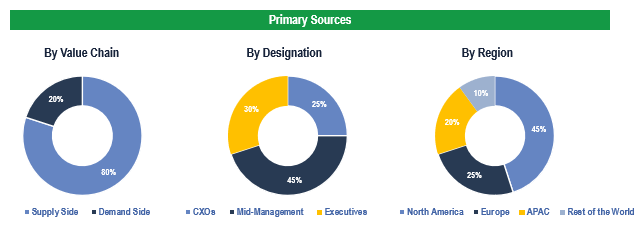
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Ambulatory Surgery Centers/Clinics, and Endoscopy Centers.
Breakdown of Primary Interviews
Market Size Estimation
The market size was derived based on extensive secondary research further validated through expert interviews.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with primary interviews and an internal knowledge repository.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

