
Endovascular Devices Market Size, Share & Growth Analysis, Size, Share and Industry Analysis Report, Regional Outlook, Growth Potential, and Regional Forecast to 2031
Global Endovascular Devices Market valued at $ 5.4 billion (2026), is expected to witness a growth rate of 5% by 2031. An increasing preference for endovascular aneurysm repair (EVAR) procedures, technological advancements in endovascular devices, growing geriatric population & corresponding rise in the prevalence of cardiovascular diseases, lifestyle factors (diet, exercise), ease & availability of endovascular procedures, and reimbursements for endovascular procedures are some of the major factors driving the market’s growth.
Endovascular surgery is an innovative procedure that is performed using minimally invasive catheter techniques on the arteries or veins. Procedures include aneurysm repair, cardiovascular disorders, peripheral vascular diseases, renal vascular diseases, deep vein thrombosis (DVT), varicose vein repair, stroke, among others. A wide variety of guidewires, catheters, crossing devices, balloons, stents, atherectomy devices, drug-coated devices, and other devices are used for endovascular treatment.
Growing Preference for Endovascular Aneurysm Repair (EVAR) Procedure Opens Growth Opportunities
An aortic aneurysm is a bulge that occurs in the wall of the aorta that carries blood from the heart to the body. It increases the risk of developing a tear in the inner layer of the wall of the aorta. An aortic aneurysm can be abdominal or thoracic. An abdominal aortic aneurysm (AAA) occurs when a portion of the abdominal aorta bulges into an aneurysm because of a weakening of the vessel wall. AAA may result in life-threatening internal bleeding upon rupture and hence patients diagnosed with AAA are treated with open surgical repair or mostly via less invasive endovascular aneurysm repair (EVAR). During the EVAR procedure, a stent graft is inserted into the aorta which protects the aorta from tearing. The EVAR is widely accepted as a standard of care for aortic aneurysms & supported by vascular surgeons due to the following benefits:
- Shorter stay in the hospital.
- Relatively less blood loss
- Faster return to routine activities
- Shorter procedure time as compared to open surgery, among others
Citing the lucrative prospects of EVAR technology, companies are launching innovative products in new markets. For instance,
- In May 2021, Endologix launched Alto® Abdominal Stent Graft System in Canada and Argentina. The launch boots the company’s global reach with the latest endovascular aneurysm repair (EVAR) technology.
“Endovascular aneurysm repair (EVAR) has emerged as a safe, and effective method of treating abdominal aortic aneurysms as it lowers the risk of life-threatening tears & ruptures. Technological advancements in EVAR have not only allowed vascular surgeons to achieve therapeutic aims but also reduced procedural risks and patient discomfort." -Director, Leading Endovascular Device Manufacturer, United States
Technological Advancements Are Driving the Endovascular Devices Market
The endovascular devices market is a technology-driven market and is marked by constant product enhancements/innovations. For instance,
- In June 2022, Medtronic India launched a fourth-generation flow diverter, Pipeline Vantage with Shield Technology for endovascular treatment of brain aneurysms.
- In December 2021, Terumo Aortic launched the first commercial case of the Aortic Balloon in the United States. The device assists physicians in the expansion of the aorta when using TREO and RELAY stent-grafts in endovascular aortic repair.
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in Endovascular Devices Market
The global endovascular devices market is marked by the presence of both established and new players. Players operating in the market adopt both organic and inorganic growth strategies such as acquisitions, and new product launches to garner market share. For instance,
- In March 2022, Cook Medical received FDA Breakthrough Device Designation for Zenith® Thoraco+ Endovascular System. The Thoraco+ is built on the strength of the proven Zenith platform and represents a next-generation endovascular graft for the treatment of thoracoabdominal aortic aneurysms. The system is indicated for the endovascular treatment of patients with thoracoabdominal aortic aneurysm.
- In September 2021, Abbott acquired Walk Vascular to bolster its endovascular product portfolio.
The endovascular devices market is expected to continue its growth in the upcoming years due to growing cases of peripheral, aortic, & venous diseases, an increase in the smoking population, growth opportunities in the APAC region, technological advancements in endovascular devices, and favourable reimbursement in key markets.
Competitive Landscape Analysis: Endovascular Devices Market
The global endovascular devices market is marked by the presence of established players such as Cordis, Medtronic, Boston Scientific, Cook Medical, Abbott, Gore Medical, Cardiovascular Systems, Inc. (CSI), Terumo, among others.
Key Strategic Questions Addressed
- What is the market size & forecast of the Endovascular Devices Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Endovascular Devices Market?
- What are the key trends defining the endovascular devices market?
- What are the major factors impacting the global market?
- What are the opportunities prevailing in the endovascular devices market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the endovascular devices market?
- What are the key strategies adopted by market players?
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
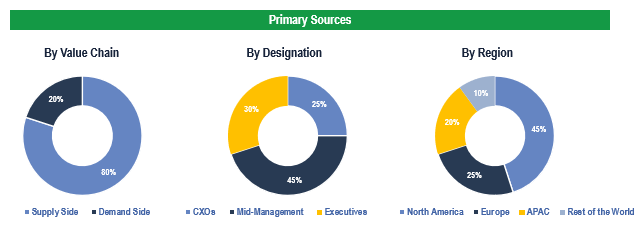
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Ambulatory Surgery Centers, Cardiac Centers, among others.
Breakdown of Primary Interviews
Market Size Estimation
The market size was derived based on extensive secondary research further validated through expert interviews.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with primary interviews and an internal knowledge repository.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

