
HPV Testing and PAP Test Market Size, Share, Analysis by Technology (Fuel Cell, Semiconductor Oxide Sensor, Others), End User (Law Enforcement Agencies, Enterprises, Individuals), Application (Alcohol Detection, Medical Applications, (Drug Abuse Detection) and Regional Forecast to 2031
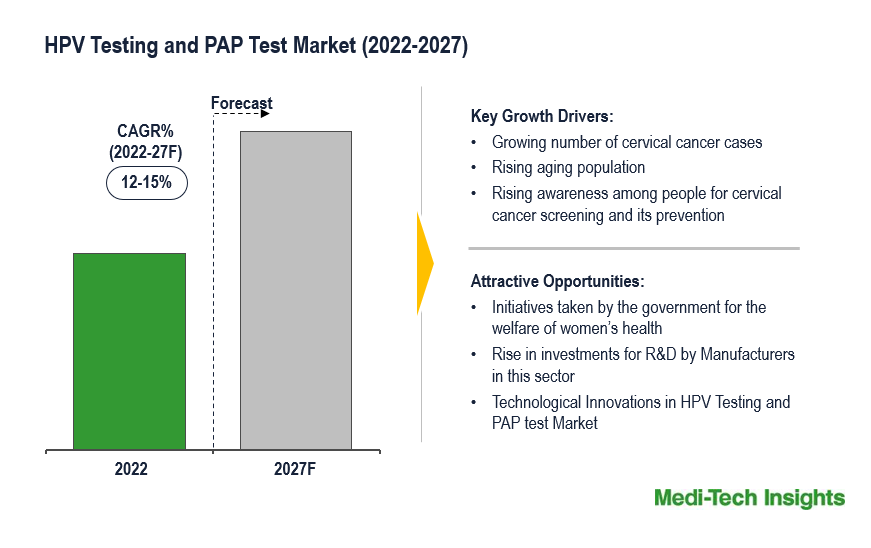
The Global HPV Testing and PAP Test Market is expected to grow at a healthy rate of 15 % by 2031. Growing number of HPV cases has led to a quick increase in the number of cervical cancers globally, and growing awareness among people for cervical cancer prevention are some of the key factors driving the market growth.
Cervical cancer is a malignant tumor that starts in the cells of the cervix, the lowermost part of the uterus. Cervical cancer starts when the cells on its surface transform into precancerous cells. If not destroyed or removed, these abnormal cells may turn into cancer cells and spread in the cervix and surrounding areas. Cervical cancer screening involves HPV and PAP tests, which can detect HPV infection or pre-cancerous lesions at an early stage and if treatment is provided at the appropriate time cancer can be prevented.
Slowdown in the HPV and PAP Test Market Owing to Covid-19 Pandemic
The Covid-19 pandemic has affected the healthcare sector in several aspects. During the pandemic due to social-distancing norms in hospitals and clinics, there was a delay in various diagnostic examinations and procedures. The delay in cancer screening services due to a shortage of staff and disruption in the supply chain of testing kits across the globe has led to an adverse impact on the HPV Testing and PAP Test market during the Covid-19 outbreak.
However, in April 2020 amidst the pandemic when all non-emergence procedures stopped, in Sweden HPV sampling kits were distributed among the eligible group of women across the country.
“When everything stopped, we had to think of something completely different. The expectation is that many women will be prevented from developing cervical cancer and that we will progress faster on the path to reaching the WHO target of elimination of cervical cancer with the help of these kits.”-Cancer Specialist, Research-Led Medical University, Sweden
Technological Advancements in HPV Testing and PAP Tests Fuels the Demand of HPV Testing and PAP Test Market
The growing demand for the development of technologically advanced screening tests for the early detection of cervical and vaginal cancers is expected to drive the HPV testing and PAP test market. The growth in the number of screening campaigns arranged by government and non-government organizations is another key factor driving the market growth across the world.
For instance,
- In Feb 2023, BD (Becton, Dickinson and Company) announced that U.S. Food and Drug Administration (FDA) approved BD Onclarity™ HPV Assay to be used with the ThinPrep® Pap Test. BD Onclarity™ HPV Assay can individually detect and identify 14 high-risk HPV genotypes in a single analysis.
- In April 2020, Roche announced that it received FDA approval for the cobas® HPV test for use on the fully automated, high-throughput cobas® 6800/8800 Systems.
Rising Cases of Cervical Cancer Boosts the Growth of HPV Testing and PAP Test Market
The growing cases of the number of cervical cancers, the rising aging population and the development of advanced technological screening tests are the key factors driving the HPV Testing and PAP Test market. Public awareness campaigns initiated by the government and other organizations, improving health insurance coverage, and favorable reimbursement policies are some of the other factors fueling the demand for the HPV Testing and PAP Test market globally.
For instance,
- In August 2022, Sansure Biotech got approval from NMPA (National Medical Products Administration China), for marketing Human Papillomavirus DNA (23 genotypes) Diagnostic Kit (PCR-Fluorescence Probing) developed using fast nucleic acid release technology and can screen 18 high-risk types and 5 low-risk types genotypes for cervical cancer.
- In June 2022, Roche announced the launch of a human papillomavirus (HPV) self-sampling solution in countries accepting the CE mark. The sampling solution allows a patient to privately collect samples for HPV screening at a healthcare facility by following instructions of a healthcare worker.
Key Market Challenges: HPV Testing and PAP Test Market
The adoption of revised regulatory guidelines for cervical cancer screening, the availability of HPV vaccination globally, and the high costs of screening tests in developing countries may hinder the growth of the global HPV Testing and PAP Test market in the upcoming years.
North America Accounts for the Largest Share of the Global HPV Testing and PAP Test Market
North America is expected to dominate the global HPV Testing and PAP Test market. This can be mainly attributed due to the growing number of cervical cancers and awareness programs, favorable reimbursement policies, the presence of key market players, and the developed healthcare system in this region. However, the Asia-Pacific region is expected to grow with the highest CAGR in the forecast period. Prime factors such as technological advancements, free screening programs, and strategic initiatives taken by public and private organizations are some of the key factors driving the Asia-Pacific market.
Competitive Landscape Analysis: HPV Testing and PAP Test Market
The key and established players operating in the global HPV Testing and PAP Test market are listed below:-
- Abbott Laboratories
- F. Hoffmann-La Roche AG
- Becton, Dickinson and Company
- Quest Diagnostics
- Hologic Inc.
- Femasys Inc.
- Arbor Vita Corporation
- Seegene Inc.
- Thermo Fischer Scientific Inc.
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the HPV Testing and PAP Test Market
All key and established players operating in this global market are adopting organic and inorganic growth strategies such as collaborations, acquisitions, and new product launches to garner a larger market share.
For instance,
- In May 2022, the London School of Hygiene & Tropical Medicine (LSHTM) in collaboration with the University of Manchester and University of Hull, initiated the ‘Catch-up Screen’ project which will offer an HPV test to women of age 65 and above by working with local GP practices. It is expected that approximately 10,000 women will be invited to participate in this project, which will help in evaluating if at-home tests are an effective way to reduce cancer in this older age group. This project has received £1.5 million in funding from the charity Yorkshire Cancer Research and will commence in April 2023.
The global HPV Testing and PAP test market is a growing market and is expected to gain a consistent momentum in the upcoming years due to a rapid surge in awareness for early diagnosis of cervical cancer, investment in R&D to introduce several advanced screening tests, and aggressive organic and inorganic growth strategies adopted by the leading market players.
Key Strategic Questions Addressed in this Research Report:
- What is the market size & forecast for the HPV Testing and PAP Test market ?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the HPV Testing and PAP Test market?
- How has Covid-19 impacted the HPV Testing and PAP Test market ?
- What are the major growth drivers, restraints/challenges impacting the global market?
- What are the opportunities prevailing in the HPV Testing and PAP Test market?
- What is the investment landscape of HPV Testing and PAP Test market ?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the HPV Testing and PAP Test market?
- What are the key strategies adopted by players in HPV Testing and PAP Test market ?
- Research Methodology
1.1. Secondary Research
1.2. Primary Research
1.3. Market Estimation
1.4. Market Forecasting - Executive Summary
- Market Overview
3.1. Market Dynamics
3.1.1. Drivers
3.1.2. Restraints
3.1.3. Opportunities
3.1.4. Market Trend
3.2. Industry Speaks
3.3. Technological Advancements - Epidemiological Assessment of Cervical Cancer and HPV
- Global HPV Testing and PAP Test Market - Size & Forecast (2026-2031), By Product
5.1. Instruments
5.2. Consumables
5.3. Services - Global HPV Testing and PAP Test Market - Size & Forecast (2026-2031), By Technology
6.1. PCR
6.2. Immunodiagnostics
6.3. Other Technologies - Global HPV Testing and PAP Test Market - Size & Forecast (2026-2031), By Application
7.1. Cervical Cancer Screening
7.2. Vaginal Cancer Screening - Global HPV Testing and PAP Test Market - Size & Forecast (2026-2031), By End-User
8.1. Hospitals
8.2. Laboratories
8.3. Other End-users - Global HPV Testing and PAP Test Market - Size & Forecast (2026-2031), By Region
9.1. North America (U.S. & Canada)
9.2. Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
9.3. Asia Pacific (China, India, Japan, Rest of Asia Pacific)
9.4. Rest of the World (Latin America, Middle East & Africa) - Competitive Landscape
10.1. Key Players and their Competitive Positioning
10.1.1. Competitive Positioning of Key Players (2026)
10.1.2. Segment-wise Player Mapping
10.2. Key Strategies Assessment, By Player (2026-2031)
10.2.1. New Product & Service Launches
10.2.2. Partnerships, Agreements, & Collaborations
10.2.3. Mergers & Acquisitions
10.2.4. Geographic Expansion - Key Companies Scanned (Indicative List)
11.1. Abbott Laboratories
11.2. F. Hoffmann-La Roche AG
11.3. Becton, Dickinson and Company
11.4. Quest Diagnostics
11.5. Hologic Inc.
11.6. Femasys Inc.
11.7. Arbor Vita Corporation
11.8. Seegene Inc.
11.9. Thermo Fischer Scientific Inc.
11.10. Other Prominent Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)

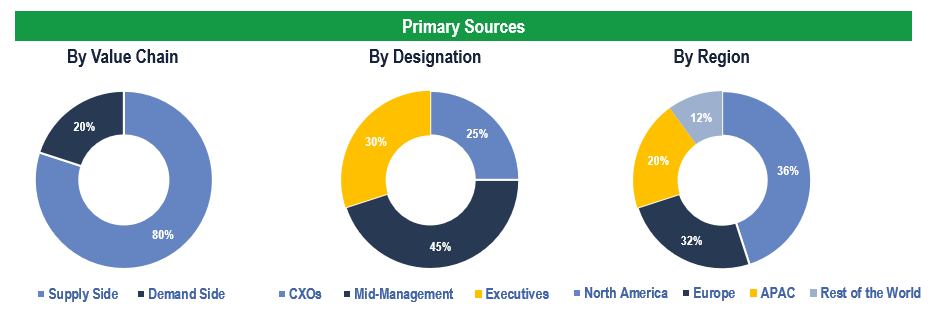
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Clinics/Laboratories, and Physician Offices.
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

