Global Life Science Consulting Services Market Size, Share, Trends, Growth Analysis and Forecasts 2024 to 2029
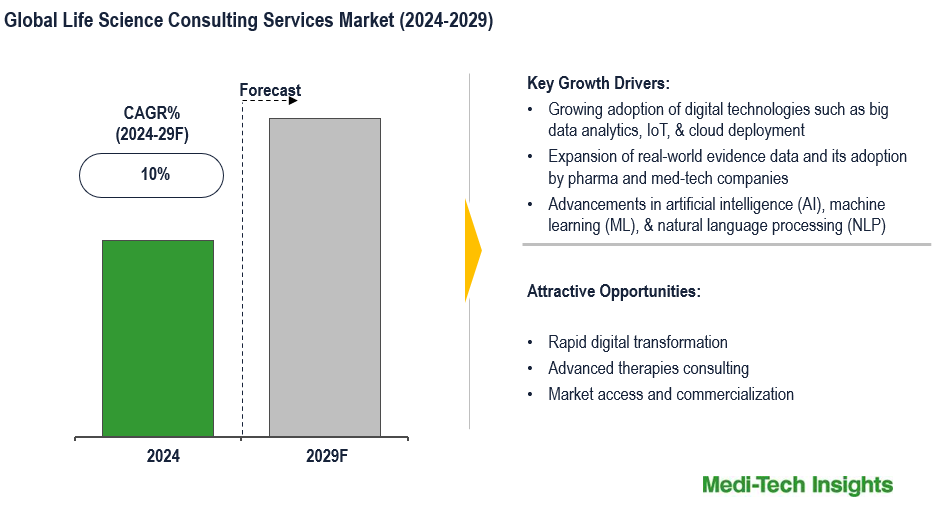
The Global Life Science Consulting Services Market is expected to witness a growth rate of 10% in the next five years. Growing adoption of digital technologies in life science; expansion of real-world evidence data and its adoption by pharma and med-tech companies; advancements in artificial intelligence (AI), machine learning (ML), & natural language processing (NLP); growing need to develop improved healthcare strategies for better outcomes; and increasing pressure to reduce rising healthcare costs are some of the key factors driving the global life science consulting services. However, concerns regarding data security and confidentiality, growing number of multi-sourcing approaches, and high cost of consulting services are some of the key challenges that are likely to hamper the market growth. To learn more about the research report, download a sample report.
Life science consulting assists companies to enhance their management and operations with regards to services such as clinical development services, regulatory services, compliance consulting, and pricing & market access, among others. These life science consulting services help pharma and med-tech firms manage their internal organizational structures more effectively, thereby delivering better outcomes.
Growing Adoption of Digital Technologies Such as Big Data Analytics, IoT, & cloud deployment to Drive Market Growth
The growing adoption of digital health technologies like big data analytics, IoT, and cloud deployment is significantly driving the demand and growth of life science consulting services. These technologies enhance data collection and analysis, enabling more informed decision-making and operational efficiency. Big Data Analytics enables the extraction of valuable insights from vast datasets, optimizing research and development processes. IoT enhances real-time monitoring and data collection from connected devices, improving patient care and operational efficiency. Cloud deployment offers scalable and cost-effective solutions for data storage and sharing, facilitating collaboration across research teams and healthcare providers. Together, these technologies streamline operations, enhance data-driven decision-making, and accelerate innovation in the life sciences sector. As organizations seek to leverage these advanced technologies, the demand for specialized consulting services that provide strategic guidance, implementation support, and compliance assurance is rising, driving the growth of life science consulting services.
“The growing adoption of digital technologies is transforming the life sciences industry, and consulting services are playing a key role in helping companies navigate this digital transformation and unlock the full potential of these technologies to improve patient outcomes and drive business success"- Director – Digital Health, A Global Leading Pharma Company, United States
Expansion of Real-World Evidence Data and its Adoption by Pharma and Med-Tech Companies to Fuel Market Growth
RWE encompasses data collected from various sources outside traditional clinical trials, such as electronic health records, insurance claims, patient registries, and wearable devices. This data provides insights into how treatments perform in real-world settings, enabling more accurate assessments of efficacy, safety, and patient outcomes. Pharmaceutical and med-tech companies are increasingly leveraging RWE to inform drug development, regulatory submissions, market access strategies, and post-market surveillance. The integration of RWE into these processes helps reduce development costs, speed up time-to-market, and improve patient-centric care. However, effectively utilizing RWE requires expertise in data analytics, regulatory requirements, and health economics.
Life science consulting services are in high demand as companies seek specialized knowledge to navigate the complexities of RWE. Consultants provide strategic guidance on data collection and integration, advanced analytics to interpret RWE, and support in regulatory compliance and submissions. They also assist in developing robust real-world evidence generation strategies that align with business objectives. As the use of RWE becomes more prevalent, the need for consulting services that can harness its full potential continues to grow, driving the expansion of the life science consulting sector.
To learn more about this report, download the PDF brochure
Demand for Advanced Therapies Consulting to Offer New Opportunities in the Life Science Consulting Services Market
Advanced therapies, such as cell and gene therapies (CGT) and personalized vaccines require specialized expertise throughout the development, manufacturing, and regulatory process. Due to the rapidly evolving Advanced Therapy Medicinal Products (ATMP) landscape, it’s crucial to have up-to-date knowledge and expertise to successfully develop a product and gain a centralized market authorization (MA) in Europe and/or a Biologic License Authorization (BLA) in the US. With rapidly expanding research on cell and gene therapies and a healthy CGT pipeline, there is robust demand for ATMP consulting as it requires experts with specific experience in regulatory strategy, CMC, medical communications, and clinical development.
Growing Demand for Market Access Services – a Key Trend in Life Science Consulting Services Market
Pharmaceutical companies are now increasingly required to demonstrate how a new drug improves patient outcomes, reduces the burden of the healthcare system as a whole, and is worth its price. To support these criteria, pharmaceutical companies are looking at a more holistic view of patient treatment, providing better RWE and therefore a stronger value proposition for decision-makers. Moreover, complex regulatory and reimbursement pathways, varied evidence requirements, and long procedural timelines pose risk to successful access and launch. All these factors are boosting the demand for market access services including Health Economics and Outcomes Research (HEOR), PV, and RWE strategy and consulting across the globe.
To learn more about this report, download the PDF brochure
Service Type Segment Analysis
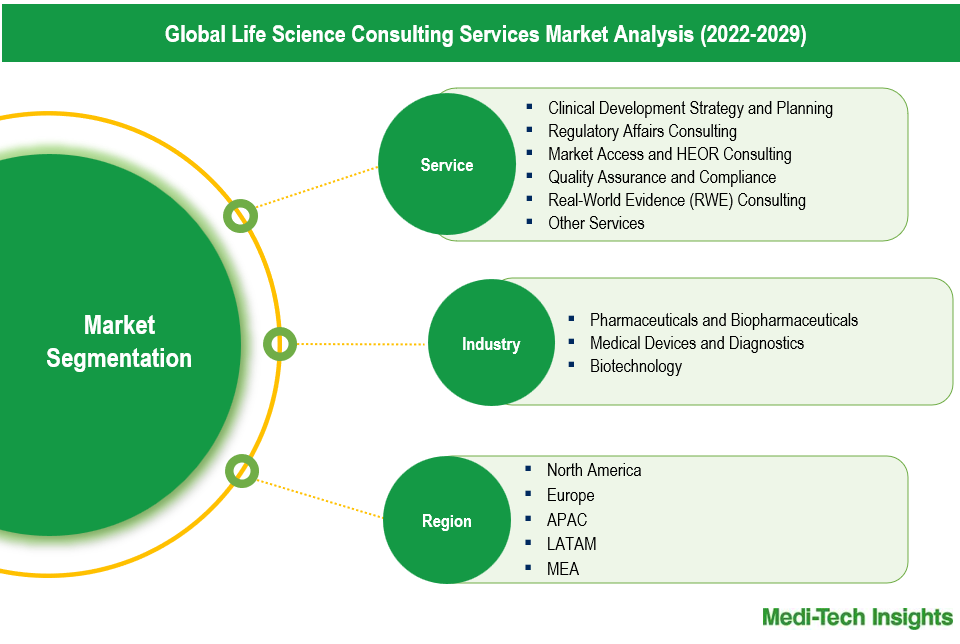
The life science consulting services market comprises service types including: Clinical Development Strategy and Planning, Regulatory Affairs Consulting, Market Access and HEOR Consulting, Quality Assurance and Compliance, Real-World Evidence (RWE) Consulting, and Other Services. Market Access and HEOR Consulting is the largest service type segment. This segment's growth is driven by pharmaceutical companies' need to demonstrate drug value through improved patient outcomes, healthcare system burden reduction, and cost-effectiveness, necessitating comprehensive HEOR and market access strategies. Additionally, RWE Consulting is the fastest-growing service type segment. The increasing importance of RWE in regulatory decision-making, reimbursement strategies, and demonstrating real-world drug effectiveness drives demand. These services provide critical insights that help pharmaceutical companies navigate complex regulatory pathways and varied evidence requirements, ensuring successful drug access and launch.
Industry Type Segment Analysis
The life science consulting services market can be analyzed based on industry types, such Pharmaceuticals and Biopharmaceuticals, Medical Devices and Diagnostics, and Biotechnology. Pharmaceuticals and Biopharmaceuticals is the largest end-user segment. This sector's size is attributed to the extensive need for consulting services across various stages of drug development, market access, regulatory affairs, and health economics. Pharmaceutical companies often require comprehensive support to navigate complex regulatory environments, optimize drug pricing, and demonstrate value through Health Economics and Outcomes Research (HEOR). The Biotechnology sector is the fastest-growing end-user segment. The rapid advancement in biotechnological innovations, coupled with increasing investment in biotech startups and R&D, drives the demand for specialized consulting services. Biotech firms often seek expertise in clinical development, regulatory strategies, and real-world evidence to accelerate their product development and market entry. The dynamic nature of biotechnological advancements contributes to the swift growth of consulting services tailored to this sector
Competitive Landscape Analysis: Life Science Consulting Services Market
The global Life Science Consulting Services market is marked by the presence of established market players such as IQVIA; Parexel; Certara; Inizio; PharmaLex GmbH; Halloran Consulting Group; Fishawack Limited; Envision Pharma Group; Uniphar Group; Science Group; Indegene; Azenta and Life Sciences among others.
Get a sample report for competitive landscape analysis
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as collaborations, acquisitions, and new product launches to garner market share. For instance,
- In June 2024, UST, a leading digital transformation solutions company, acquired Endeavor Consulting Group, a consulting firm specializing in life sciences operations to enhance its capabilities in supply chain management and product commercialization, particularly within the life sciences sector. This acquisition will boost UST's technological and operational expertise, improving client efficiency and innovation in the pharmaceutical, biopharma, and medical technology industries
- In March 2024, Infosys acquired life sciences consulting and technology leader, BASE Life Science, enhancing its capabilities in cloud-based digital platforms and data solutions for the life sciences sector. This move expands Infosys' presence in the Nordics and strengthens its expertise in clinical trials, drug development, and data-driven health outcomes
- In October 2023, EVERSANA acquired Healthware Group, a full-service agency and innovation consultancy, enhancing its European operations and global capabilities in commercial services for the life sciences industry. The merger integrates Healthware’s expertise with EVERSANA INTOUCH, expanding their reach and innovation in product launches, market access, and stakeholder engagement
- In February 2023, Accenture acquired Bionest, a consulting firm specializing in complex biopharma strategies, enhancing its capabilities in precision medicine, oncology, and cell & gene therapies. This acquisition will bolster Accenture's Life Sciences practice with Bionest’s expertise and expand its go-to-market strategies
- In November 2022, Lumanity acquired Clarion Healthcare, enhancing its global consulting practice with Clarion's expertise in commercialization and product lifecycle management. This acquisition strengthens Lumanity’s capabilities in developing and executing complex strategies for biopharma clients
The life science consulting services market is expected to gain further momentum in the coming years due to technological advancements, rising R&D investments, new service launches, and aggressive organic and inorganic growth strategies followed by the players.
Future Outlook of the Life Science Consulting Services Market
The global Life Science Consulting Services market is expected to gain further momentum in the coming years due to the complex regulatory requirements for drug approval and market access, the need for specialized expertise in emerging areas such as personalized medicine, and integration of advanced technologies. These factors collectively contribute to the growth and evolution of the life science consulting services market.
Life Science Consulting Services Market Report Scope
| Report Scope | Details |
| Base Year Considered | 2023 |
| Historical Data | 2022 - 2023 |
| Forecast Period | 2024 - 2029 |
| CAGR (2024-2029) | 10% |
| Segment Scope | Product, Application, End User |
| Regional Scope |
|
| Key Companies Mapped | IQVIA; Parexel; Certara; Inizio; PharmaLex GmbH; Halloran Consulting Group; Fishawack Limited; Envision Pharma Group; Uniphar Group; Science Group; Indegene; Azenta and Life Sciences among others |
| Report Highlights | Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Key Strategic Questions Addressed
-
What is the market size & forecast for the Global Life Science Consulting Services Market?
-
What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global Life Science Consulting Services Market?
-
How has COVID-19 impacted the Global Life Science Consulting Services Market?
-
What are the major growth drivers, restraints/challenges impacting the market?
-
What are the opportunities prevailing in the market?
-
What is the investment landscape?
-
Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
-
Who are the major players operating in the market? What is the competitive positioning of key players?
-
Who are the new players entering the market?
-
What are the key strategies adopted by players?
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
-
- Market Dynamics
- Drivers
- Restraints
- Key Market Trends
- Industry Speaks
- Market Dynamics
- Key Revenue Pockets
- Global Life Science Consulting Services Market - Size & Forecast (2022-2029), By Service Type
- Clinical Development Strategy and Planning
- Regulatory Affairs Consulting
- Market Access and HEOR Consulting
- Quality Assurance and Compliance
- Real-World Evidence (RWE) Consulting
- Other Services
- Global Life Science Consulting Services Market - Size & Forecast (2022-2029), By Industry Type
- Pharmaceuticals and Biopharmaceuticals
- Medical Devices and Diagnostics
- Biotechnology
- Global Life Science Consulting Services Market - Size & Forecast (2022-2029), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Competitive Positioning of Key Players (2023)
- Offerings Assessment, By Player
- Key Strategies Assessment, By Player (2022-2024)
- New Product & Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Key Companies Scanned (Indicative List)
- IQVIA
- Parexel
- Certara
- Inizio
- PharmaLex GmbH
- Halloran Consulting Group
- Fishawack Limited
- Envision Pharma Group
- Uniphar Group
- Science Group
- Indegene
- Azenta Life Sciences
- Other Prominent Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
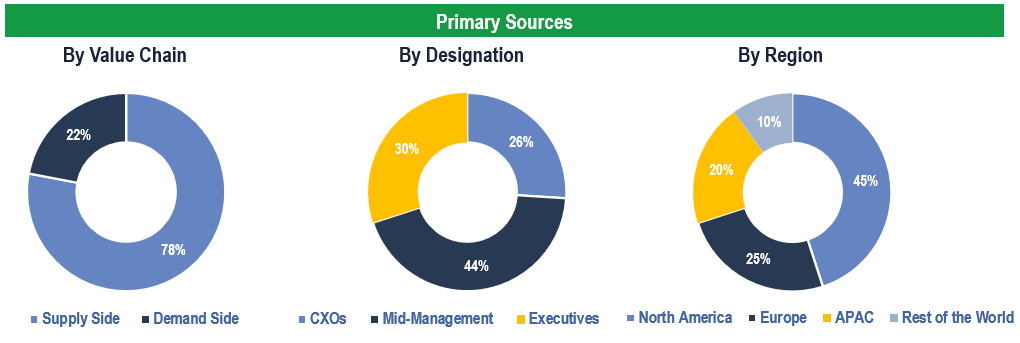
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Pharma, Biotech, CROs, CDMOs & Other End Users
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel