
Nerve Repair and Regeneration Market Size, Share, Trend Overview Segmented By Product(Biomaterials, Nerve Conduits, Nerve Wraps), Application(Nerve Grafting, Stem Cell Therapy), Region, Leading Key Players and Forecast To 2031
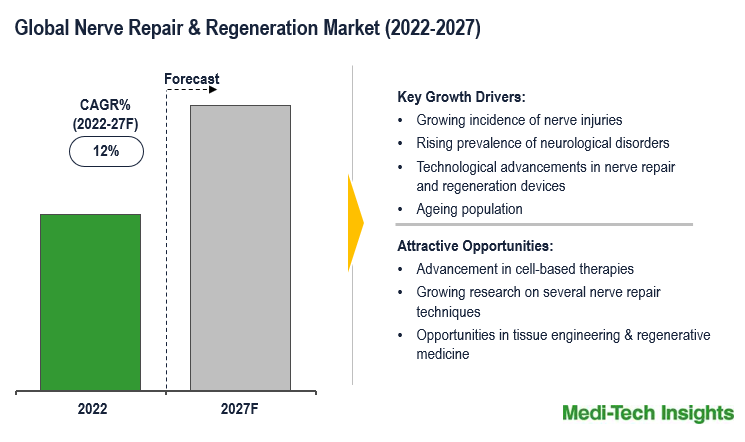
The Global Nerve Repair and Regeneration Market is expected to grow at a rate of ~12% by 2031. Growing cases of nerve injuries, rising number of neurological disorders, technological advancements in nerve repair and regeneration devices, growing geriatric population, and growing investments by the government to support research activities in neurological disorders are some of the key factors driving the market growth.
Nerves are bundles of fibers that carry messages between the brain and the rest of the body. Nerves run all over the body and can be injured, resulting in problems with sensation or movement. Repair and regeneration in the nervous system is a process by which damaged tissue undergoes regrowth or renewal, eventually restoring nervous system function. This process happens more readily with the peripheral nervous system axons, synapses, neurons, and glia.
Technological Advancements in Neurostimulation & Neuromodulation Devices Boosts the Growth of Nerve Repair and Regeneration Market
Neurostimulation & Neuromodulation devices and treatments are life-changing. They affect every area of the body and treat nearly every disease or symptom from headaches to tremors to spinal cord damage, and urinary incontinence. These devices find their usage in many applications such as to treat traumatic or neurodegenerative disorders such as Alzheimer's disease, amyotrophic lateral sclerosis, multiple sclerosis, multiple system atrophy, deep brain stimulation (DBS) treatment for Parkinson's disease, sacral nerve stimulation for pelvic disorders and incontinence, and spinal cord stimulation for ischemic disorders (angina, peripheral vascular disease). The adoption of neurostimulation & neuromodulation devices in various applications is contributing to the growth of the nerve repair and regeneration market.
For instance,
- In December 2022, Abbott announced that they have received the U.S. Food and Drug Administration (FDA) approval for the company's Eterna spinal cord stimulation (SCS) system for the treatment of chronic pain. The neuromodulation device provides an optimized experience with the ability to wirelessly charge as few as five times per year, the lowest recharge burden compared to other rechargeable SCS systems.
- In March 2022, MyndTec announced that they have received FDA approval for MyndMove 2.0, its second-generation neuromodulation MyndMove system for clinical and home usage.
- In January 2022, Medtronic announced that they have received U.S. Food and Drug Administration approval of its Intellis rechargeable neurostimulator and Vanta recharge-free neurostimulator for the treatment of chronic pain associated with diabetic peripheral neuropathy (DPN).
Growing Prevalence of Neurological Disorders Drives the Nerve Repair & Regeneration Market
Neurological disorders are a growing and serious problem in many parts of the world. According to the WHO, more than 1 billion people throughout the world are affected by some type of neurological disease. Nearly 6 million people die every year as a result of a neurological disorder. The specific causes of neurological problems vary but can include genetic disorders, congenital abnormalities or disorders, infections, lifestyle or environmental health problems including malnutrition, brain injury, spinal cord injury, or nerve injury. With a growing population and aging society, more people are reaching ages at which neurological diseases are highly prevalent, stroke is the leading cause of disability and mortality, resulting in enormous human and economic burdens. The rising burden of neurological disorders among the population is likely to increase the demand of nerve repair & regeneration market.
Stem Cell-Based Therapies Opens Up New Growth Opportunities in Nerve Repair and Regeneration Market
Peripheral nerve injuries (PNIs) are some of the most common types of traumatic lesions affecting the nervous system. Although the peripheral nervous system has a higher regenerative ability than the central nervous system, delayed treatment is associated with disturbances in both distal sensory and functional abilities. The research findings suggest that the pluripotential stem cell from bone marrow, adipose tissue, skin, dental pulp, umbilical cord, or from nerve trunks can differentiate into Schwann cells and nerve cells with axons which could further help in the regeneration of the missing nerves. The function of these cells is supposed to be a combination of self-renewal function, production, and release of many growth factors, preparing the environment for nerve growth and providing a closed space for axons to grow. Furthermore, the use of stem cells derived from different sources has presented promising results for nerve regeneration and effective neural and functional results which in turn has further boosted the nerve repair and regeneration market growth.
Key Constraints/ Challenges: Nerve Repair and Regeneration Market
The high costs of the repair and regeneration devices, the dearth of skilled professionals, difficulty in treating large nerve gaps, and the growing preference for using drug therapies over nerve repair & regeneration products are some of the factors restraining the growth of the nerve repair and regeneration market in the upcoming years.
North America Expected to Continue to Hold a Major Share in the Nerve Repair and Regeneration Market
From a geographical perspective, North America holds a major market share of the global nerve repair and regeneration market. This can be mainly attributed to the rising geriatric population, growing adoption of technologically advanced products, presence of key market players, and favorable reimbursement landscape in the region. However, the Asia-Pacific region is expected to witness a strong growth in the upcoming years due to an increase in the aging population, growing demand for nerve repair and regenerative therapies, government initiatives to support research activities in neurological disorders, and the constantly improving healthcare infrastructure in the region.
Competitive Landscape Analysis: Nerve Repair and Regeneration Market
Some of the leading players operating in the global nerve repair and regeneration market are listed below:-
- AxoGen Inc.
- Stryker Corporation
- Baxter International Inc.
- Polyganics B.V.
- Boston Scientific Inc.
- Phageneis
- Integra Lifesciences Holding Corporation
- Abbott Laboratories
- Cyberonics Inc.
- Nevro Corp
- NeuroPace Inc.
- Medtronic plc., among others.
Organic Growth Strategies Adopted by the Market Players to Establish Their Foothold in the Nerve Repair and Regeneration Market
All players operating in the nerve repair & regeneration market are adopting organic and inorganic growth strategies such as collaborations, acquisitions, and new product launches to garner a larger market share.
For instance,
- In January 2023, Abbott announced that they have received the U.S. Food and Drug Administration (FDA) approval for its Proclaim XR spinal cord stimulation (SCS) system to treat painful diabetic peripheral neuropathy (DPN), a debilitating complication of diabetes.
- In October 2022, Phageneis announced that they have received United States FDA de novo approval for the Phagenyx neurostimulation system, which uses pharyngeal electrical stimulation to restore swallowing control in patients with severe dysphagia post-stroke.
- In April 2022, Boston Scientific announced that they have received FDA approval for Boston Scientific’s STIMVIEW XT guided programming software, which allows clinicians to visualize in real-time both lead placement and stimulation modeling of brain anatomy for patients with Parkinson’s disease (PD) or essential tremor who undergo deep brain stimulation (DBS).
The nerve repair and regeneration market is a growing market that is expected to gain further momentum in the coming years due to technological advancements in enabling technologies, new product launches, and aggressive organic growth strategies followed by the market players.
Key Strategic Questions Addressed in this Report
- What is the market size & forecast for the nerve repair and regeneration market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the nerve repair and regeneration market?
- How has Covid-19 impacted the nerve repair and regeneration market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the nerve repair and regeneration market?
- What is the investment landscape of nerve repair and regeneration market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the nerve repair and regeneration market?
- What are the key strategies adopted by players working in nerve repair and regeneration market?
1. Research Methodology
1.1. Secondary Research
1.2. Primary Research
1.3. Market Estimation
1.4. Market Forecasting
2. Executive Summary
3. Market Overview
3.1. Market Dynamics
3.1.1. Drivers
3.1.2. Restraints
3.1.3. Opportunities
3.1.4. Market Trend
3.2. Industry Speaks
3.3. Technology Trends
4. COVID-19 Impact on Nerve Repair & Regeneration Market
5. Reimbursement Landscape: Snapshot
6. Global Nerve Repair & Regeneration Market- Size & Forecast (2026-2031), By Product
6.1. Neurostimulation & Neuromodulation Devices
6.1.1. Internal Neurostimulator devices
6.1.2. External Stimulator devices
6.2. Biomaterials
6.2.1. Nerve Conduits
6.2.2. Nerve Wraps
7. Global Nerve Repair & Regeneration Market- Size & Forecast (2026-2031), By Application
7.1. Neurostimulation & Neuromodulation Surgeries
7.2. Direct Nerve Repair/Neurorrhaphy
7.3. Nerve Grafting
7.4. Stem Cell Therapy
8. Global Nerve Repair & Regeneration Market- Size & Forecast (2026-2031), By Region
8.1. North America (U.S. & Canada)
8.2. Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
8.3. Asia Pacific (China, India, Japan, Rest of Asia Pacific)
8.4. Rest of the World (Latin America, Middle East & Africa)
9. Competitive Landscape
9.1. Key Players and their Competitive Positioning
9.1.1. Market Share Analysis (2022)
9.1.2. Segment-wise Player Mapping
9.2. Key Strategies Assessment, By Player (2020-2022)
9.2.1. New Product & Service Launches
9.2.2. Partnerships, Agreements, & Collaborations
9.2.3. Mergers & Acquisitions
9.2.4. Geographic Expansion
10. Key Companies Scanned (Indicative List)
10.1. AxoGen Inc.
10.2. Stryker Corporation
10.3. Baxter International Inc.
10.4. Polyganics B.V.
10.5. Boston Scientific Inc.
10.6. Phageneis
10.7. Integra Lifesciences Corporation
10.8. Cyberonics Inc.
10.9. Medtronic plc.
10.10. Abbott Laboratories
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Ambulatory Surgery Centers, Clinics, and Other End Users.
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

