
Pacemakers Market (2026-2031) Size, Share, Analysis By Product (Implantable Pacemakers, External Pacemakers), Technology (Single-chamber pacemakers, Dual-chamber pacemakers), Application (Conventional Pacemakers, MRI Compatible Pacemakers), End Use, Region and Segment Forecast
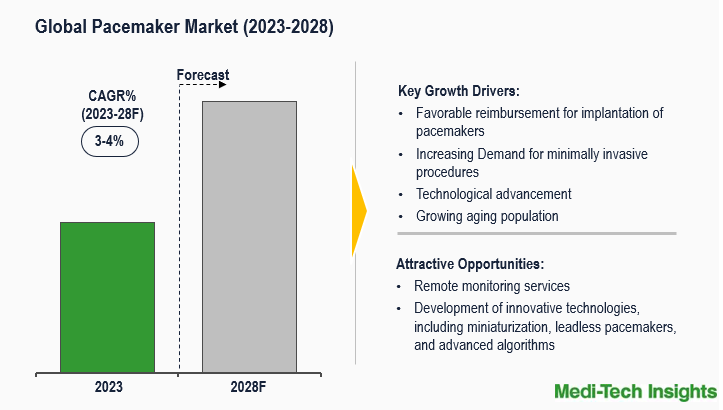
The Pacemakers Market is expected to grow at a rate of 3-4% by 2031. The key factors driving the market are the increasing prevalence of cardiovascular diseases, the growing demand for minimally invasive procedures, advancements in pacemaker technology and minimally invasive procedures, the increasing aging population, and favorable reimbursement in the key markets. To learn more about the research report, fill out a quick inquiry for a sample report.
Pacemakers are medical devices that regulate the heart's rhythm by sending electrical impulses to the heart muscles. They treat various heart conditions, such as arrhythmias and bradycardia. Pacemakers are broadly classified into the following three main types:
- Single chamber: This type involves a single lead attached to either the upper (atrium) or lower (ventricle) chamber of the heart. It is suitable for patients with specific rhythm disturbances that can be addressed by stimulating only one chamber
- Dual-chamber: Dual-chamber pacemakers employ two leads, one for the upper (atrium) and one for the lower (ventricle) heart chambers. This design allows for a more synchronized and natural pacing of the heart, closely mimicking its physiological rhythm
- Biventricular Pacemakers: Biventricular pacemakers are utilized in cardiac resynchronization therapy (CRT) to address heart failure. These pacemakers have three leads: one in the right atrium, one in the right ventricle, and an additional lead in the left ventricle
Advancements in Pacemaker Technology Drives Market Growth
The outlook for pacemakers is promising, thanks to ongoing advancements in microelectronics and battery technology. This progression has led to the creation of more compact devices with added functionalities. Notably, enhancements in both battery capacity and technological capabilities, coupled with imaginative designs, are expected to extend the longevity of pacemakers. Anticipate the incorporation of innovative features designed to streamline follow-up procedures, ensuring efficiency for both patients and clinical staff. Moreover, these innovations are poised to revolutionize real-time monitoring of heart activity, heralding an era where adjustments to therapy delivery are automated. The integration of these breakthroughs is set to enhance the effectiveness and convenience of pacemaker usage in the years ahead and reduce the need for invasive procedures. For Instance,
- In August 2023, BIOTRONIK announced the full market release of its newest family of pacemaker and CRT-P devices, with patient-centric technologies for better patient care and workflow simplification
- In May 2023, Medtronic plc, a global leader in healthcare technology, announced that they have received U.S. Food and Drug Administration (FDA) approval of its Micra™ AV2 and Micra™ VR2, the next generation of its industry-leading miniaturized, leadless pacemakers
Fill out the "Quick Inquiry Form" to request a sample copy.
Escalating Cardiovascular Challenges Spur the Adoption of Pacemakers Globally
The upsurge in cardiac issues is driving hospitalizations and medical procedures, leading to increased pacemaker adoption. Behavioral risk factors, such as an unhealthy diet, reduced physical activity, heightened tobacco use, and excessive alcohol consumption, contribute to the rising prevalence and mortality rates of cardiovascular diseases. Additionally, genetic factors and an aging population also play a role. According to the Institute for Health Metrics and Evaluation (IHME), the total cases of cardiovascular diseases surged from 271 million in 1990 to over 520 million in 2019, with associated mortalities rising from 12.1 million to 18.6 million during the same period. Thus, the increasing severity of cardiovascular issues globally and the examination of how pacemakers are being embraced and utilized on a worldwide scale to address these challenges are significantly driving the market growth.
North America leads the global market and is poised to sustain its dominance
From a geographical perspective, North America holds a major market share of the pacemaker's market. This growth is primarily attributed to the region's well-established healthcare industry. The presence of key industry players, a rising elderly population facing cardiac issues, rapid technological advancements like MRI-compatible pacemakers and Bluetooth-enabled pacemakers, and improving regulatory approvals are significant factors contributing to the ongoing expansion of the regional market. According to the American Heart Association, nearly half of the US population grapples with heart-related concerns, driving the demand for advanced cardiovascular equipment and medical interventions.
Pacemakers Market: Competitive Landscape
Some of the key players operating in the market include Boston Scientific, Cook Medical, GE Healthcare, ZOLL Medical Corporation, Medtronic PLC, LivaNova PLC, Osypka Medical GmbH, Biotronik SE & Co. KG, Lepu Medical Co. Ltd, and Abbott Laboratories, among others.
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market Players
Players operating in this market are adopting organic and inorganic growth strategies such as collaborations, and acquisitions to garner market share. For instance,
- In December 2023, BIOTRONIK announced the opening of its new Asia Pacific Manufacturing and Research Hub. The 20,000 m² site will serve as the company’s central hub in Asia-Pacific, with hundreds of employees working in manufacturing, quality, research & development (R&D), sales and marketing for implantable pacemakers, defibrillators, and vascular intervention
- In July 2023, Abbott announced that the U.S. Food and Drug Administration (FDA) has approved the AVEIR dual chamber (DR) leadless pacemaker system, the world's first dual chamber leadless pacing system that treats people with abnormal or slow heart rhythms
The market is further expected to grow due to the launch of new products, advancement in pacemaker technologies, the growing integration of remote monitoring features in pacemakers, and aggressive organic and inorganic growth strategies followed by the players.
Pacemakers Market Scope
|
Report Metric |
Details |
|
Market Growth Rate |
3-4% by 2028 |
|
Historical data |
2019 – 2023 |
|
Base year |
2023 |
|
Forecast period |
2023-2028 |
|
Key Driving Factors |
|
|
Pacemaker Types |
|
|
Recent Technological Developments |
|
|
Competitive Landscape |
Key Players: Boston Scientific, Cook Medical, GE Healthcare, ZOLL Medical Corporation, Medtronic PLC, LivaNova PLC, Osypka Medical GmbH, Biotronik SE & Co. KG, Lepu Medical Co. Ltd, Abbott Laboratories, among others |
|
Growth Strategies of Key Players |
|
|
Future Growth Factors |
|
Key Strategic Questions Addressed
- What is the market size and forecast for the Pacemakers Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Pacemakers Market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the market?
- What is the investment landscape?
- Which region has the highest share in the market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the market?
- What are the key strategies adopted by players?
- Research Methodology
-
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Industry Speaks
- Market Dynamics
- Global Pacemakers Market - Size & Forecast (2026-2031), By Product
- Implantable Pacemakers
- External Pacemakers
- Global Pacemakers Market - Size & Forecast (2026-2031), By Technology
- Single-chamber pacemakers
- Single-chamber atrial
- Single-chamber ventricular
- Dual-chamber pacemakers
- Biventricular/CRT pacemakers
- Single-chamber pacemakers
- Global Pacemakers Market - Size & Forecast (2026-2031), By Application
- Conventional Pacemakers
- MRI Compatible Pacemakers
- Global Pacemakers Market - Size & Forecast (2026-2031), By End-use
- Hospitals
- Cardiac Care Centre
- Ambulatory Surgical Centers
- Clinics
- Others
- Global Pacemakers Market - Size & Forecast (2026-2031), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and Their Competitive Positioning
- Competitive Positioning of Key Players (2022)
- Offerings Assessment, By Players
- Key Strategies Assessment, By Player (2026-2031)
- New Product Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Companies Scanned (Indicative List)
- Boston Scientific
- Lepu Medical Co. Ltd
- Cook Medical
- GE Healthcare
- Abbott Laboratories
- Medtronic PLC
- Biotronik SE & Co. KG
- ZOLL Medical Corporation
- LivaNova PLC
- Osypka Medical GmbH
- Other Prominent Players
- Key Players and Their Competitive Positioning
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
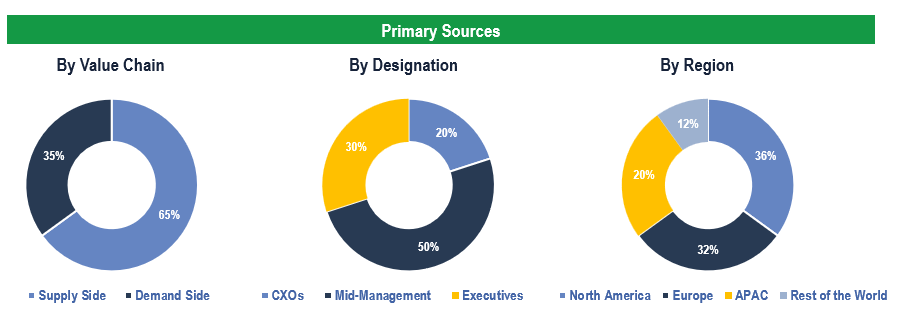
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
- Demand Side Stakeholders: Hospitals, Clinics, Ambulatory Centers, and Cardiac Catheterization Laboratories, among others
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & and internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Frequently Asked Questions (FAQs)
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

