Global Real-world Evidence (RWE) Solutions Market Size, Growth & Trends Report Segmented by Component (Services, Data Sets), Application (Drug Development & Approvals, Medical Device Development & Approvals), End-user, & Regional Forecast to 2030
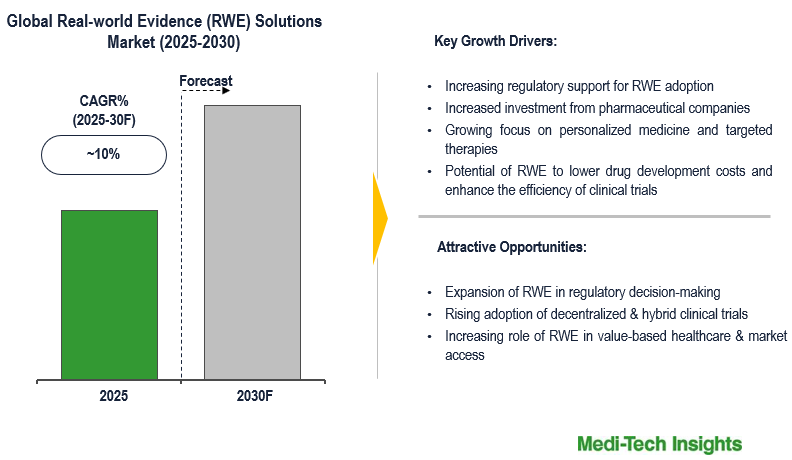
The real-world evidence (RWE) solutions market is expected to expand at a CAGR of ~10% during the forecast period. Key factors driving this growth include increasing regulatory support for RWE adoption, the rising incidence of chronic diseases, increased investment from pharmaceutical companies, the growing focus on personalized medicine and targeted therapies, the widespread adoption of electronic health records (EHRs) and digital health data, and the potential of RWE to lower drug development costs and enhance the efficiency of clinical trials. To learn more about the research report, download a sample report.
Report Overview
Real-world evidence (RWE) solutions refer to advanced analytical tools, platforms, and methodologies that leverage real-world data (RWD) to generate clinical insights on the usage, effectiveness, safety, and value of medical products, treatments, and healthcare interventions. These solutions integrate data from various sources including electronic health records (EHRs), claims databases, patient registries, wearable devices, and patient-reported outcomes to support drug development, regulatory decision-making, market access, post-market surveillance, and value-based healthcare initiatives. RWE solutions help pharmaceutical companies, healthcare providers, payers, and regulatory bodies assess treatment outcomes, improve patient care, optimize clinical practices, and inform reimbursement and policy decisions. Real-world evidence (RWE) has been in use for decades, but recent advances in digital technology and advanced analytics allow it to be employed in new ways.
To learn more about this report, download the PDF brochure
RWE plays a vital role in helping regulators formulate decisions
RWE could potentially help the FDA while making regulatory decisions about the safety and effectiveness of medical products such as identifying new safety issues post-approval or determining the effectiveness of a drug for a new indication or patient population. Unlike traditional clinical trials, which are conducted in controlled environments with limited patient populations, RWE provides insights from diverse patient groups, routine clinical practice, and post-market surveillance. This enables regulators to evaluate treatment outcomes, monitor adverse events, and refine drug labeling based on real-world usage. Additionally, RWE supports expedited approvals and label expansions by supplementing clinical trial data, especially in areas like rare diseases, personalized medicine, and oncology. Citing the potential, the 21st Century Cures Act (Cures Act, signed in 2016) required the FDA to publish a framework for a program to evaluate the use of RWE in regulatory decision-making; FDA published the framework for this program in December 2018. As part of its RWE Program, the FDA committed to understanding the full potential of RWD and RWE in regulatory decision-making. The agency has been holding workshops, funding demonstration projects, creating mobile and web applications that can collect RWD, and engaging with sponsors on the topic.
Pharma/biopharma companies are focusing on strengthening RWE capabilities
The ability to rapidly convert real-world data sources, such as claims data and electronic medical records (EMRs), into actionable evidence enhances patient health outcomes by enabling pharmaceutical companies to streamline drug development and optimize commercialization strategies. These insights empower biopharmaceutical firms to accelerate the development of effective therapies, generate robust evidence for payers, and strengthen their competitive positioning in the healthcare market. To access high-quality real-world data and refine study designs, many pharma companies are establishing strategic collaborations with healthcare providers, payers, and data analytics firms. Enhancing RWE capabilities also allows them to adapt to the growing focus on value-based care and reimbursement models, ensuring that new treatments align with both regulatory and payer expectations. Consequently, the industry is shifting toward integrated RWE platforms, real-time evidence generation, and adaptive trial designs, reinforcing RWE as a key driver of innovation and improved patient outcomes.
To learn more about this report, download the PDF brochure
Competitive Landscape Analysis
The global real-world evidence (RWE) solutions market is marked by the presence of established and emerging market players such as IQVIA Holdings; Parexel; Icon; PPD; Syneos Health; Clarivate; Cerner; Medpace Holdings; Flatiron Health; Symphony Innovation; Aetion; Cognizant Technology; IBM; Oracle; Data Matrix; SAS Institute Inc.; Clinigen Group; and UnitedHealth Group; among others. Some of the key strategies adopted by market players include new product development, strategic partnerships and collaborations, and investments.
Report Scope
| Report Metric | Details |
| Base Year Considered | 2024 |
| Historical Data | 2023 - 2024 |
| Forecast Period | 2025 - 2030 |
| Growth Rate | ~10% |
| Market Drivers |
|
| Attractive Opportunities |
|
| Segment Scope | Component, Application, and End User |
| Regional Scope |
|
| Key Companies Mapped | IQVIA Holdings; Parexel; Icon; PPD; Syneos Health; Clarivate; Cerner; Medpace Holdings; Flatiron Health; Symphony Innovation; Aetion; Cognizant Technology; IBM; Oracle; Data Matrix; SAS Institute Inc.; Clinigen Group; and UnitedHealth Group |
| Report Highlights | Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Global Real-world Evidence (RWE) Solutions Market Segmentation
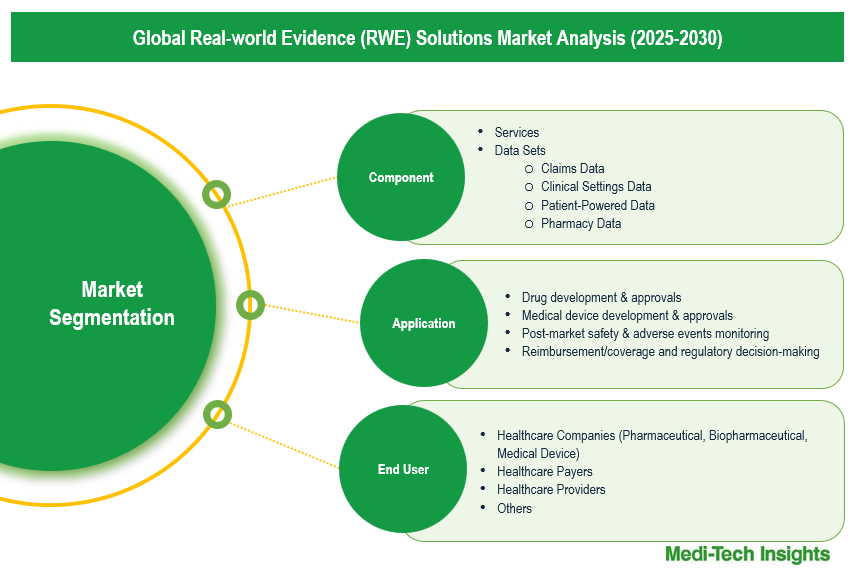
This report by Medi-Tech Insights provides the size of the global real-world evidence (RWE) solutions market at the regional- and country-level from 2023 to 2030. The report further segments the market based on component, application, and end user.
Market Size & Forecast (2023-2030), By Component, USD Million
- Services
- Data Sets
- Claims Data
- Clinical Settings Data
- Patient-Powered Data
- Pharmacy Data
Market Size & Forecast (2023-2030), By Application, USD Million
- Drug development & approvals
- Medical device development & approvals
- Post-market safety & adverse events monitoring
- Reimbursement/coverage and regulatory decision making
Market Size & Forecast (2023-2030), By End User, USD Million
- Healthcare Companies (Pharmaceutical, Biopharmaceutical, Medical Devices)
- Healthcare Payers
- Healthcare Providers
- Others
Market Size & Forecast (2023-2030), By Region, USD Million
- North America
- US
- Canada
- Europe
- UK
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Middle East & Africa
Key Strategic Questions Addressed
- What is the market size & forecast of the real-world evidence (RWE) solutions market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the real-world evidence (RWE) solutions market?
- What are the key trends defining the market?
- What are the major factors impacting the market?
- What are the opportunities prevailing in the market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market?
- What are the key strategies adopted by players?
- Introduction
- Introduction
- Market Scope
- Market Definition
- Segments Covered
- Regional Segmentation
- Research Timeframe
- Currency Considered
- Study Limitations
- Stakeholders
- List of Abbreviations
- Key Conferences and Events (2025-2026)
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Bottom-Up Approach
- Top-Down Approach
- Market Forecasting
- Executive Summary
- Real-world Evidence (RWE) Solutions Market Snapshot (2025-2030)
- Segment Overview
- Regional Snapshot
- Competitive Insights
- Market Overview
- Market Dynamics
- Drivers
- Increasing regulatory support for RWE adoption
- Rising incidence of chronic diseases
- Increased investment from pharmaceutical companies
- Growing focus on personalized medicine and targeted therapies
- Adoption of electronic health records (EHRs) and digital health data
- Potential of RWE to lower drug development costs and enhance the efficiency of clinical trials
- Restraints
- Interoperability issues with legacy systems
- Data inconsistency and quality issues
- Data security and privacy concerns
- Opportunities
- Expansion of RWE in regulatory decision-making
- Rising adoption of decentralized & hybrid clinical trials
- Increasing role of RWE in value-based healthcare & market access
- Key Market Trends
- Integration of genomics & RWE for precision medicine
- Growing adoption of blockchain for the secure health data exchange
- Collaboration between pharma, tech, & data companies
- Unmet Market Needs
- Industry Speaks
- Global Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Component, USD Million
- Introduction
- Services
- Data Sets
- Claims data
- Clinical settings data
- Patient-powered data
- Pharmacy data
- Global Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Application, USD Million
- Introduction
- Drug Development & Approvals
- Medical Device Development & Approvals
- Post-Market Safety & Adverse Events Monitoring
- Reimbursement/Coverage And Regulatory Decision Making
- Global Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By End User, USD Million
- Introduction
- Healthcare Companies (Pharmaceutical, Biopharmaceutical, Medical Devices)
- Healthcare Payers
- Healthcare Providers
- Others
- Global Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Region, USD Million
- Introduction
- North America Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Country, USD Million
- US
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Canada
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- US
- Europe Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Country, USD Million
- UK
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Germany
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- France
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Italy
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Spain
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Rest of Europe
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- UK
- Asia Pacific (APAC) Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), By Country, USD Million
- China
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Japan
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- India
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Rest of Asia Pacific
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- China
- Latin America (LATAM) Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), USD Million
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Middle East & Africa (MEA) Real-world Evidence (RWE) Solutions Market Size & Forecast (2023-2030), USD Million
- Market Size & Forecast, By Component (USD Million)
- Market Size & Forecast, By Application (USD Million)
- Market Size & Forecast, By End User (USD Million)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Key Player Comparison
- Segment-wise Player Mapping
- Market Share Analysis (2024)
- Company Categorization Matrix
- Dominants/Leaders
- New Entrants
- Emerging Players
- Innovative Players
- Key Strategies Assessment, By Player (2022-2024)
- New Product Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Company Profiles*
(Business Overview, Financial Performance**, Products Offered, Recent Developments)
- Oracle Corporation
- IBM Corporation
- IQVIA Holdings
- Parexel
- Icon
- PPD
- Syneos Health
- Clarivate
- Cerner
- Data Matrix
- Other Prominent Players
Note: *Indicative list
**For listed companies
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
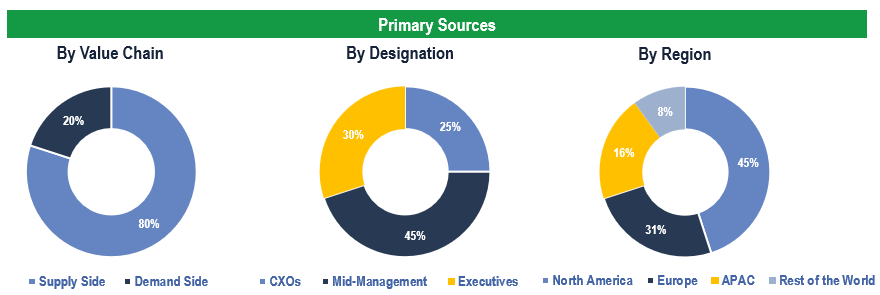
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders from Healthcare Companies (Pharmaceutical, Biopharmaceutical, Medical Devices), Healthcare Payers, Healthcare Providers, and Others
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down & Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, the Internal Knowledge Repository, and the Company’s Sales Data.