Spinal Muscular Atrophy Treatment Market – Awareness on SMA Diagnosis & Treatments
The Global Spinal Muscular Atrophy Treatment Market is expected to grow at a rate of 13% by 2026. The rising prevalence of spinal muscular atrophy (SMA), growing awareness regarding spinal muscular atrophy (SMA) diagnosis and treatment, rising number of initiatives to improve treatment options for rare diseases, and increasing R&D activities to develop novel therapies & treatments for spinal muscular atrophy (SMA) are some of the key factors driving the global spinal muscular atrophy treatment market. However, the high cost associated with the treatment of spinal muscular atrophy (SMA) is likely to hamper the market growth.
Spinal Muscular Atrophy refers to a group of hereditary diseases that damages and kills specialized nerve cells in the brain and spinal cord (called motor neurons). Motor neurons control movement in the arms, legs, face, chest, throat, and tongue, as well as skeletal muscle activity including speaking, walking, swallowing, and breathing. There are four types of SMA:
- Type 1 is the most common and severe form of SMA. It’s sometimes called Werdnig-Hoffmann disease or infantile-onset SMA and is usually evident before 6 months of age.
- Type 2 is an intermediate form of SMA. Usually first noticed between 6 and 18 months of age. Children can sit without support but are unable to stand or walk unaided.
- Type 3 is a milder form of SMA. It’s also known as Kugelberg-Welander disease and resembles muscular dystrophy that can be seen after the age of 18 months. Children can walk independently but may have difficulty walking or running, rising from a chair, or climbing stairs.
- Type 4 is very rare. It usually starts in young adulthood and causes mild motor impairment.
Increasing R&D Activities for the Treatment of Spinal Muscular Atrophy Drives the Spinal Muscular Atrophy Treatment Market
Currently, there is no known cure for Spinal Muscular Atrophy (SMA). However, multiple treatments are available to help slow the progression of the disease, relieve symptoms, and manage potential complications. In addition to these approved treatments, several other treatments are being tested in clinical trials. For instance,
- In January 2022, Cytokinetics announced that their collaboration with Astellas to advance the development of CK-2127107/ reldesemtiv ended in late 2021.
- In August 2021, Novartis announced the lift of a partial clinical trial hold and planned to initiate a new, pivotal Phase 3 study of intrathecal OAV-101 in older patients with spinal muscular atrophy.
- In January 2021, Biogen Inc. announced that they have treated their first patient in the global clinical study, RESPOND. The Phase 4 study examines the clinical benefit and assesses the safety of SPINRAZA® (Nusinersen) in infants and children with spinal muscular atrophy (SMA) who still have unmet clinical needs following treatment with gene therapy.
Growing Adoption of Spinal Muscular Atrophy Treatment in Babies Fuels the SMA Treatment Market Demand
Spinal muscular atrophy affects approximately 1 in 11,000 babies born worldwide each year and is the most common genetic cause of death in infants. There are several types of SMA that affect children. They vary in the age when symptoms first appear, the seriousness of symptoms, and how symptoms get worse. Early detection and treatment of SMA are important as studies suggest that therapy is most effective when started in the first few months of life., For children, treatment includes targeted therapy, respiratory therapy, nutritional therapy, muscle & joint therapy, and medications depending on the symptoms. Several innovative drugs have recently been developed that improve or ameliorate symptoms in many children.
North America Holds the Largest Market Share in Spinal Muscular Atrophy Treatment Market
North America holds the largest market share of the spinal muscular atrophy treatment market followed by Europe and the Asia Pacific. The surge in the demand for drugs for the treatment including Nusinersen, Onasemnogene & Abeparvovec, growing healthcare expenditure, favorable reimbursement policies for the treatment of rare diseases, growing awareness regarding SMA, and a rise in the number of R&D activities for the treatment of SMA in this region are some of the key factor driving the growth of the global spinal muscular atrophy treatment market.
Competitive Landscape Analysis: Spinal Muscular Atrophy Treatment Market
The spinal muscular atrophy treatment market is marked by the presence of established market players such as Astellas Pharma Inc., AstraZeneca PLC, Biogen Inc., Boehringer Ingelheim GmbH, Catalyst Pharmaceuticals, Chugai Pharmaceutical, Cytokinetics Inc., F. Hoffmann-La Roche AG, Cure SMA, PTC Therapeutics Inc., Novartis AG, Ionis Pharmaceuticals, NMD Pharma A/S, Genentech Inc, Avexis Inc., Pfizer Inc., Regeneron Pharmaceuticals, Abbott, Novo Nordisk A/S, Takeda Pharmaceutical Company Limited, and Salarius Pharmaceuticals Inc., among others.
Companies Adopt both Organic & Inorganic Growth Strategies to Increase their Market Share in SMA Treatment Market
Leading players operating in this market are adopting both organic and inorganic growth strategies such as collaborations, acquisitions, and expansions to garner a higher market share. For instance,
- In May 2022, PTC Therapeutics Inc. announced that they have received U.S. Food and Drug Administration (FDA) approval for a label extension for Evrysdi® (risdiplam) to include infants under 2 months old with spinal muscular atrophy (SMA).
- In August 2021, Cytokinetics and Cure SMA announced the renewal of their partnership to increase education, awareness, public policy, and fundraising for spinal muscular atrophy (SMA).
- In May 2021, Biogen and Ginkgo Bioworks announced the collaboration & license agreement to develop novel gene therapy manufacturing platform to treat certain neurological and neuromuscular diseases as well as other conditions across multiple therapeutic areas.
The spinal muscular atrophy treatment market is a growing market that is expected to gain further momentum in the coming years due to a growing number of companies with pipeline candidates in advanced stages of a clinical trial, emerging treatment therapies in the treatment of SMA, a rise in the number of research and development activities for improving the therapies for SMA, and aggressive organic and inorganic growth strategies followed by the market players.
Key Strategic Questions Addressed
- What is the market size & forecast of the spinal muscular atrophy treatment market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the spinal muscular atrophy treatment market?
- How has Covid-19 impacted the spinal muscular atrophy treatment market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the spinal muscular atrophy treatment market?
- What is the investment landscape of spinal muscular atrophy treatment market?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the spinal muscular atrophy treatment market?
- What are the key strategies adopted by players operating in spinal muscular atrophy treatment market?
The study has been compiled based on the extensive primary and secondary research.
Secondary Research (Indicative List)
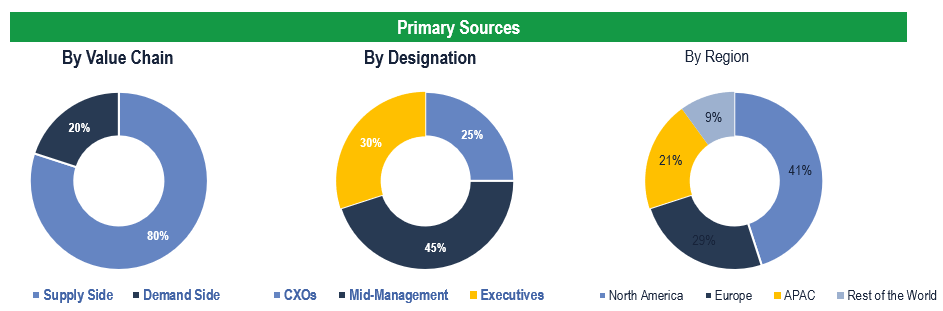
Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Specialty Clinics, Homecare, and Other End Users.
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.