
3D Cell Culture Market Report (2026-2031) Size, Share, Analysis, By Technology (Scaffold-based 3D Cell Culture, Microfluidics-based 3D Cell Culture), Application (Cancer Research, Stem Cell Research, Toxicity Testing), End User (Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Cosmetic Industry) Region and Segment Forecast
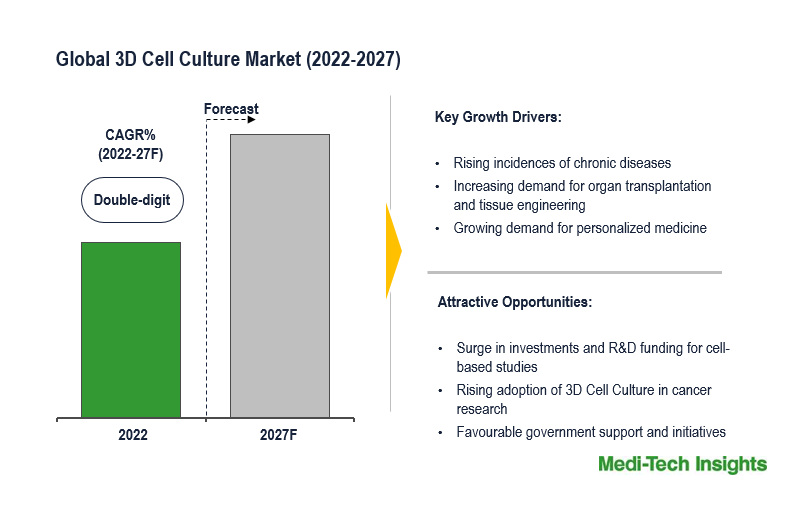
The Global 3D Cell Culture Market is expected to witness a double-digit growth rate by 2031. Some of the key factors driving the growth of the global 3D cell culture market are increasing demand for organ transplants and tissue engineering coupled with advancements in scaffold-free technology, a surge in investments and research and development (R&D) funding for cell-based studies, rising efforts to develop potential alternatives to animal-based testing, rising incidence of chronic diseases, among others. To learn more about the research, fill out a quick inquiry for a sample report.
3D cell culture is a vital technique in cell biology and tissue engineering that involves growing and studying cells within a three-dimensional environment, mimicking the conditions found in the human body. It offers numerous advantages, including the ability to replicate in vivo conditions, support the growth of complex multicellular structures, and provide realistic scenarios for drug testing. Various methods are used for 3D cell culture, such as scaffold-based approaches utilizing materials like hydrogels. Its applications encompass cancer research, tissue engineering, and drug development, leading to more accurate assessments of drug effects. However, it presents challenges, including increased complexity, the need for specialized equipment and expertise, and potentially higher costs compared to 2D cell culture. Bioprinting is often integrated into 3D cell culture, enabling precise cell and biomaterial placement, while organoids, a specific type of 3D cell culture, have gained prominence in disease modeling and drug testing.
Advancing 3D Cell Culture Techniques: Meeting the Demands of Modern Research
3D cell culture is a technique gaining traction in cell biology and tissue engineering due to several compelling factors. First and foremost is the demand for more physiologically relevant cell culture models. Traditional 2D cultures often fail to replicate in vivo conditions accurately, leading to inaccurate results in drug testing, disease modeling, and toxicity studies. Improved drug testing is another driving force, as 3D cell culture provides a superior platform for predicting drug behaviour within the human body, potentially reducing drug development costs and timelines.
Tissue engineering benefits greatly from 3D cell culture, enabling the creation of complex tissues and organs for transplantation and research. In cancer research, 3D cell cultures offer more accurate tumour growth and behaviour modeling compared to traditional monolayer cultures. Furthermore, stem cell research benefits from 3D culture systems that support stem cell differentiation and growth.
For instance,
- In June 2023, Vertex Pharmaceuticals Incorporated and Lonza unveiled a strategic partnership aimed at facilitating the production of Vertex's range of prospective stem cell-derived, fully specialized insulin-producing islet cell therapies designed for individuals with Type 1 Diabetes (T1D). The collaboration primarily focuses on the VX-880 and VX-264 programs, both of which are presently undergoing clinical trials
- In October 2022, Corning Incorporated expanded its 3D cell culture offerings by introducing the Elplasia12K flask to its product line. This flask is specifically designed to facilitate spheroid culture, a method commonly utilized in cancer research and the advancement of therapeutic treatments.
The rise of personalized medicine allows the creation of 3D cell culture models using a patient's cells for customized drug testing and treatment plans. Innovations in biomaterials, such as hydrogels and scaffolds, simplify the creation of 3D cell culture environments. Interdisciplinary collaboration among researchers further propels the development of 3D cell culture techniques.
For instance,
- In June 2023, Bristol Myers Squibb received approval from the U.S. Food and Drug Administration (FDA) for commercial production at their recently established cell therapy manufacturing facility in Devens, Massachusetts. This facility is a vital part of Bristol Myers Squibb's growing global network for producing cell therapies, ensuring a consistent supply of these treatments in the long term, and delivering these therapies to patients worldwide who have unmet medical needs
- In March 2021, Thermo Fisher Scientific introduced the Gibco Human Plasma-like Medium (HPLM), marking the debut of the first cell culture medium that replicates the metabolic characteristics of human plasma. This innovative medium is specifically created to offer researchers a more authentic perspective on cell growth as it occurs within the human body.
Revolutionary Trends in Cell Culture: Organoids and Microfluidics as Catalysts
The utilization of organoids in 3D cell cultures, which more closely mimics natural tissue architecture than traditional 2D cell cultures, is a significant trend for various reasons. It allows for disease modeling, particularly in conditions like cancer, neurodegenerative disorders, and gastrointestinal ailments, aiding in the study of disease progression and treatment testing. Additionally, organoids are increasingly crucial in drug testing and development, offering more accurate assessments of drug efficacy and safety. Furthermore, they enable personalized medicine by using a patient's own cells, resulting in more effective, tailored treatments. Organoids also contribute to understanding tissue development and physiology, shedding light on normal organ function and development. Notably, the use of organoids and microtissues in 3D cell culture has the potential to reduce ethical concerns by decreasing the reliance on animal testing in some cases. This trend is expected to continue to grow as technology and methodologies for working with these cellular models evolve.
For instance,
- In July 2023, Thermo Fisher Scientific announced the introduction of the Gibco™ OncoPro™ Tumoroid Culture Medium Kit. It is the first commercially available culture medium specifically designed for the growth of patient-derived tumoroids, also known as cancer organoids, from various cancer types. Thermo Fisher's innovative modular kit is aimed at making complex cancer models more accessible to researchers, addressing these challenges and facilitating advancements in cancer research
- In February 2023, Corning Incorporated unveiled its most recent advancements in automation and 3D cell culture during the annual Society for Laboratory Automation and Screening (SLAS) conference held in San Diego, California. Within this context, Corning Life Sciences introduced a comprehensive array of tools for 3D culture, featuring the innovative Elplasia® 12K flask and an upcoming Elplasia plate designed with an open-well format to simplify the manipulation of spheroids and organoids.
Microfluidic 3D cell culture is an advanced technique used in cell biology and tissue engineering. Microfluidics involves the manipulation of small volumes of fluids in micro-scale channels. It has gained prominence in cell culture due to its ability to precisely control the microenvironment of cells. Microfluidic-based 3D cell cultures provide a better approximation of the in vivo cellular environment, making them useful for drug testing and disease modeling. Microfluidic systems can be designed for high-throughput screening of compounds, making them valuable in drug development. Researchers can control factors like nutrient supply, oxygen concentration, and waste removal with precision in these systems.
Key Constraints/Challenges
The global 3D cell culture market faces numerous hurdles, including the lack of standardized protocols for result comparison, higher costs associated with specialized equipment, and the complexity of adopting new techniques. Scaling up for large-scale production and ensuring biocompatibility with biomaterials presents additional challenges, along with the need for consistent and reproducible results. Integrating 3D cell culture into high-throughput screening and complex data analysis poses technical challenges. Addressing these challenges is crucial for the continued development and expansion of 3D cell culture applications in various fields, including drug discovery, regenerative medicine, and disease modeling.
Fill out the "Quick Inquiry Form" to request a sample copy
North America Accounts for the Largest Share of the Global Market
The size of the market varies across regions, with typically more well-established services in developed countries. North America takes the lead followed by Europe and other regions. North America's market's driving factors include advanced healthcare infrastructure, presence of key players, supportive regulatory frameworks, government backing for 3D culture model development, and rise in R&D activities by the number of research institutes and universities are also expected to boost the market.
The Asia Pacific region is anticipated to experience the swiftest growth during the forecast period, driven by a rising number of chronic diseases, a thriving biotechnology sector, increased company investments due to lower manufacturing costs, growing demand for cellular therapies, and robust research potential.
Competitive Landscape
Some prominent players competing in the global 3D cell culture market include Thermo Fisher Scientific, Inc., Merck KGaA, Lonza, Corning Incorporated, Avantor, Inc., PromoCell GmbH, Tecan Trading AG, REPROCELL Inc., CN Bio Innovations Ltd, and Lena Biosciences among others.
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as enhancing manufacturing capabilities, collaborations, and acquisitions to garner market share. For instance,
- In July 2023, Merck announced a €23 million investment to expand its cell culture media production in Kansas, USA. The expansion includes the addition of 9,100 square meters of lab space and improved production capabilities
- In November 2022, FUJIFILM Corporation announced a substantial investment of $188 million to establish a cell culture media manufacturing facility in Research Triangle Park (RTP), North Carolina, USA. The facility will encompass more than 250,000 square feet on 64 acres to meet the growing market demand and focus on the production of animal component-free, dry powder, and liquid media to support bioproduction and advanced therapies, contributing to the advancement of life science and healthcare research and development.
The market is expected to continue to grow due to increased demand for personalized medicine, rising adoption of 3D cell cultures in cancer research, the need for more accurate and physiologically relevant models in various fields of research and application, technological innovations, and aggressive organic and inorganic growth strategies followed by the players.
3D Cell Culture Market Report Scope
|
Report Metric |
Details |
|
Market Growth Rate |
Expected double-digit growth rate by 2027 |
|
Key Growth Drivers |
Increasing demand for organ transplants and tissue engineering Advancements in scaffold-free technology Surge in investments and R&D funding for cell-based studies Rising efforts to develop alternatives to animal-based testing Increasing incidence of chronic diseases |
|
3D Cell Culture Applications |
Applications in cancer research, tissue engineering, and drug development |
|
Trending Innovations |
Use of organoids and microfluidics in 3D cell culture Thermo Fisher's OncoPro Tumoroid Culture Medium Kit for cancer organoids Corning's advancements in automation and 3D cell culture tools |
|
Competitive Landscape |
Prominent players include Thermo Fisher Scientific, Merck KGaA, Lonza, Corning, Avantor, and others |
|
Growth Strategies |
Organic and inorganic strategies include collaborations, acquisitions, and capacity expansions Examples include Merck's investment in cell culture media production, and FUJIFILM's facility in RTP, USA |
Key Strategic Questions Addressed
- What is the market size & forecast for the Global 3D Cell Culture Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global 3D Cell Culture Market?
- How has COVID-19 impacted the Global 3D Cell Culture Market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the market?
- What is the investment landscape?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the market?
- What are the key strategies adopted by players?
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
-
- Market Dynamics
- Drivers
- Restraints
- Key Market Trends
- Industry Speaks
- Market Dynamics
- Key Revenue Pockets
- Global 3D Cell Culture Market - Size & Forecast (2026-2031), By Technology
- Scaffold-based 3D Cell Culture
- Scaffold Free 3D Cell Culture
- Microfluidics-based 3D Cell Culture
- Others
- Global 3D Cell Culture Market - Size & Forecast (2026-2031), By Application
- Cancer Research
- Stem Cell Research & Tissue Engineering
- Drug Development & Toxicity Testing
- Others
- Global 3D Cell Culture Market - Size & Forecast (2026-2031), By End User
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Cosmetic Industry
- Others
- Global 3D Cell Culture Market - Size & Forecast (2026-2031), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Competitive Positioning of Key Players (2022)
- Offerings Assessment, By Player
- Key Strategies Assessment, By Player (2021-2023)
- New Product & Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Key Companies Scanned (Indicative List)
- Thermo Fisher Scientific, Inc.
- Merck KGaA
- Lonza
- Corning Incorporated
- Avantor, Inc.
- PromoCell GmbH
- Tecan Trading AG
- REPROCELL Inc.
- CN Bio Innovations Ltd
- Lena Biosciences
- Other Prominent Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
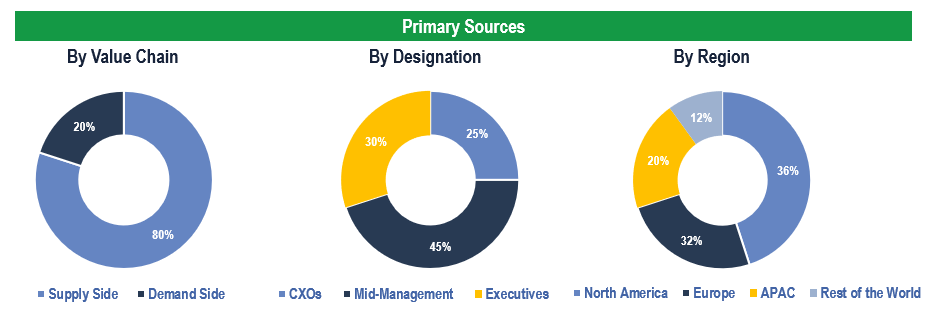
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Pharmaceutical & Biotechnology Companies, Academics & Research Institutes, and Other End Users
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel

