Non-Invasive Prenatal Testing (NIPT) Market – Global Industry Analysis, Size, Share, Trends & Growth Opportunities for Forecast 2024 to 2029
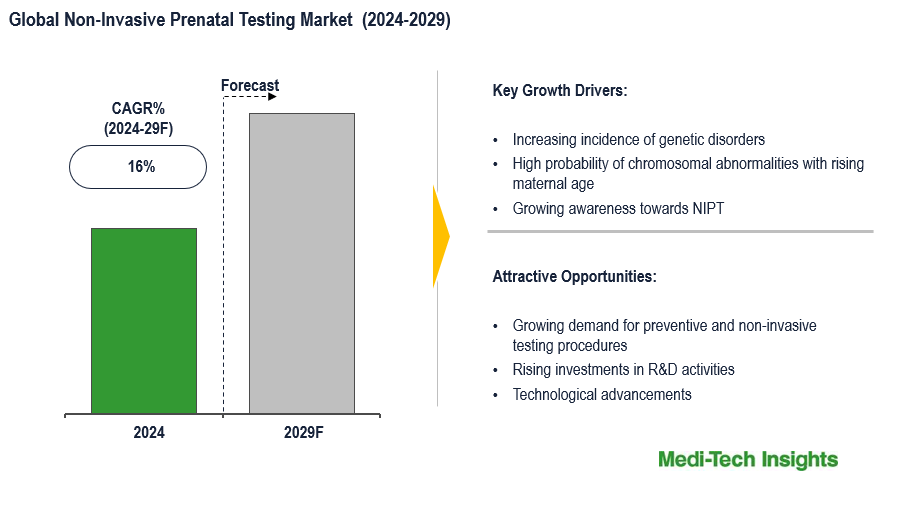
The Global Non-Invasive Prenatal Testing (NIPT) Market is expected to grow at 16% by 2029. The high risk of chromosomal abnormalities with rising maternal age, the surge in the adoption of cell-free DNA screening, favorable reimbursement policies for average and low-risk pregnancies, and a growing number of genetic and congenital disorders are some of the key factors driving the market growth. To learn more about the research report, download a sample report.
Non-invasive prenatal testing (NIPT), sometimes called noninvasive prenatal screening (NIPS), is a method of determining the risk of whether the fetus of the mother will be born with certain genetic abnormalities. This testing also analyzes small fragments of Deoxyribonucleic acid (DNA) that are circulating in a pregnant woman’s blood. NIPT is a safe and highly effective way of screening for conditions such as Down syndrome (also called trisomy 21), Edwards syndrome (trisomy 18), Patau syndrome (trisomy 13), monosomy X, and Turner syndrome.
Currently, two types of NIPT methods are commercially available, massive parallel sequencing (MPS) technology and the single-nucleotide polymorphism (SNP) based method. Both NIPT methods screen for fetal genetic disorders that arise from extra or missing chromosomes, such as trisomy 21, trisomy 18, and trisomy 13, along with fetal sex and sex chromosome disorders. NIPT can also screen conditions where a small part of the chromosome is missing, called a microdeletion, such as Di-George, Cri-du-chat, Prader-Willi, or Angelman syndrome. Approximately, 1 in 150 live births is affected by a chromosomal abnormality worldwide.
To learn more about this report, download the PDF brochure
Shift towards Cell-Free DNA screening
The Discovery of cell-free DNA (cfDNA)-based non-invasive prenatal testing (NIPT) has rapidly shifted the paradigm of aneuploidy screening during pregnancy. It’s a simple blood test that can be performed during pregnancy. Clinical validation studies suggest that NIPT is more accurate than combined first-trimester screening (CFTS), with very high sensitivity (99.3%) and specificity (99.9%) for trisomy 21(Down syndrome). For instance,
NIPT is also safer than other invasive diagnostic techniques such as chorionic villus sampling and amniocentesis, which involves a risk of miscarriage (0.1–0.2%). However, NIPT is not a diagnostic test; it can only determine whether there is an increased risk of having a baby with an abnormality. Therefore, a high-risk result should be followed by invasive testing for confirmation. Furthermore, owing to its ability to detect the presence of chromosomal abnormalities in fetuses of pregnant women as early as 9 to 10 weeks of gestation, the demand for non-invasive prenatal testing is witnessing a rapid increase globally.
Advent of Advanced Technologies for NIPT Products
Prenatal diagnosis and prenatal screening have undergone rapid development in recent years. The advent of next-generation sequencing technology has enabled the sequencing of fetal DNA fragments that can be assembled into a full genetic map, allowing the fetal genome to be scanned prenatally and non-invasively. On the other hand, the inclusion of chromosomal microarray analysis (CMA) has allowed the detection of cryptic genomic imbalances (which were not identified by previous techniques), varying levels of mosaicism, and cases of uniparental disomy (UPD). Developments in modern molecular technologies and the discovery of cell-free fetal DNA in maternal plasma have also led to novel screening methods for fetal chromosomal aneuploidies.
Currently, various types of NIPT tests are available in the market such as Panorama, Vistara, MaterniT GENOME, MaterniT 21 PLUS, and Harmony Test, among others which help in the screening of chromosomal abnormalities developing in the fetus. Moreover, several developments have been made to impact the quality of care for expectant mothers such as:
- In December 2022, Natera, Inc. disclosed in Prenatal Diagnosis a clinical experience study in twin pregnancies showcasing its Panorama single nucleotide polymorphism (SNP)-based non-invasive prenatal test (NIPT), demonstrating consistent real-world performance in line with previous reports and emphasizing the significance of Panorama's capacity to ascertain zygosity for risk evaluations
- In January 2022, QIAGEN entered into collaborations with Atila BioSystems to provide non-invasive prenatal testing (NIPT) solutions to QIAGEN’s dPCR franchise
- In June 2021, Illumina and Next-generation Genomic Thailand announced the launch of next-generation sequencing (NGS)-based VeriSeq NIPT Solution v2 in the country which helps to detect anomalies missed by targeted assays
- In December 2020, Natera announced expanded coverage from the largest U.S. Health Plan, Aetna, for its non-invasive prenatal testing (NIPT) for all pregnancies
- In June 2019, Cradle Genomics, a San Diego-based startup secured US$17 Mn in Series A funding to support clinical development, CLIA lab operations, corporate infrastructure, and commercial launch of the tests
“Prenatal care is a dynamic and continuously evolving field. Development in new molecular technologies and the discovery of cell-free fetal DNA are fueling groundbreaking advancements to improve care for mother and child and to offer parents reproductive options.” - Senior Director, Head of Research & Product Development, Sequencing & Array-based Company, United States
Key Challenges/ Constraints: Non-Invasive Prenatal Testing Market
Some of the key challenges restricting the growth of the non-invasive prenatal testing market are stringent government regulations and limitations of NIPT despite its high accuracy, it is still considered a screening test and false positive and negative results can still occur. NIPT only focuses on the common trisomies and cannot test for other genetic diseases. Various professional committees believe that the implementation of NIPT and the subsequent rise in the uptake of prenatal testing is likely to increase the incidence of abortions.
North America: The Largest Non-Invasive Prenatal Testing Market
North America is the largest market for NIPT with >45% revenue share, followed by Europe. The growing prevalence of genetic disorders in newborns associated with increasing maternal age, wider adoption and rising awareness about NIPT among the people, the introduction of new technologically advanced tests, and the presence of leading players working on fetal and neonatal care in this region are some of the key factors driving the North America market.
The market in Europe is expected to grow significantly over the forecast period due to the adoption of the latest technology, new product launches, increased awareness regarding NIPT, and a strong reimbursement framework for these tests in the region. The Asia Pacific region is expected to witness the highest CAGR in the coming years, as there is an increasing demand for early diagnosis of chromosomal disorders during pregnancies in emerging countries. Also, key market players are expanding their foothold in the emerging Asia-Pacific countries by engaging in partnerships and collaborations with domestic market players due to the presence of a large patient base in the region.
To learn more about this report, download the PDF brochure
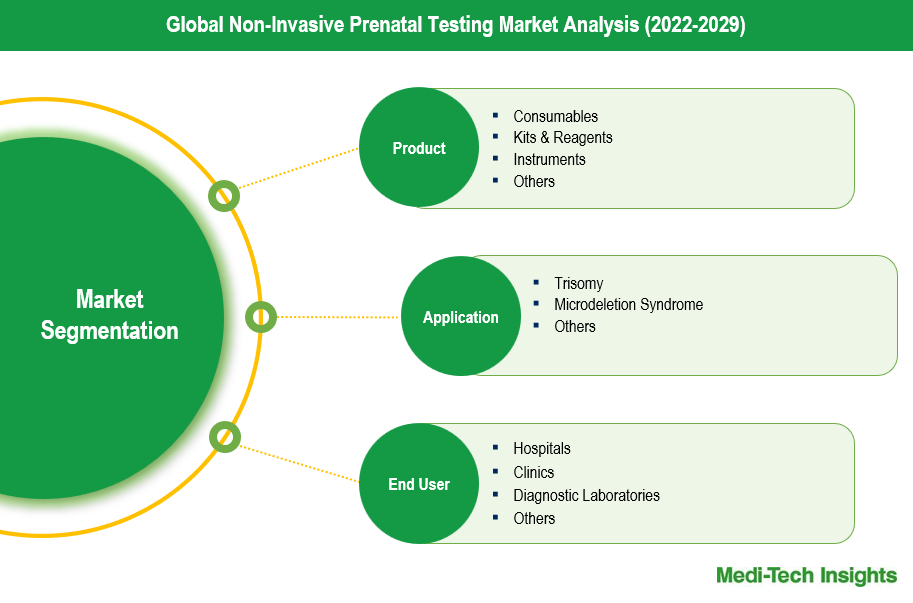
Product Segment Insights
The non-invasive prenatal testing (NIPT) market is segmented into consumables, kits & reagents, and instruments, each playing a crucial role in the overall market share. Consumables, including blood collection tubes and other materials necessary for sample preparation, hold a significant portion of the market due to their recurring usage in every test. Kits & reagents are another critical segment, encompassing the essential components for the detection and analysis of fetal genetic material from maternal blood. This sub-segment is driven by the continuous advancements in genetic testing technologies and the increasing demand for high-accuracy diagnostic tools. Instruments, such as sequencing systems and PCR machines, also constitute a substantial market share, as they are indispensable for the precise analysis and interpretation of NIPT results. The growing adoption of these sophisticated instruments in diagnostic laboratories and hospitals contributes to their strong market presence.
Application Segment Insights
The application segment of the non-invasive prenatal testing (NIPT) market is prominently categorized into trisomy and microdeletion syndrome testing, each significantly influencing market share. Trisomy testing, which includes screening for conditions such as Trisomy 21 (Down syndrome), Trisomy 18 (Edwards syndrome), and Trisomy 13 (Patau syndrome), commands the largest market share due to the high prevalence and clinical importance of these chromosomal abnormalities. The widespread adoption of trisomy NIPT is driven by its high accuracy, early detection capabilities, and the increasing preference for non-invasive methods over traditional invasive procedures like amniocentesis. On the other hand, testing for microdeletion syndromes, which involve smaller chromosomal deletions that can lead to conditions such as DiGeorge syndrome and Cri-du-chat syndrome, is rapidly gaining traction. Although currently a smaller segment compared to trisomy testing, the market share for microdeletion syndrome NIPT is expanding due to advancements in genomic technologies that enhance detection sensitivity and accuracy. Additionally, the growing awareness among healthcare providers and expectant parents about the availability and benefits of microdeletion testing is contributing to its increasing adoption, thereby steadily boosting its market share.
Competitive Landscape Analysis: Non-Invasive Prenatal Testing Market
Some of the prominent players operating in this market are Agilent Technologies, Inc., PerkinElmer, Inc., Illumina, Inc., Eurofins LifeCodexx AG, Natera, Inc., Igenomix SL, F. Hoffman-La Roche Ltd., BGI Genomics, Quest Diagnostics, Berry Genomics, and Yourgene Health among others.
Get a sample report for competitive landscape analysis
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as collaborations, acquisitions, and new product launches to garner market share. For instance,
- In May 2024, Natera introduced a new cfDNA-based fetal RhD NIPT test, aiding OB/GYN physicians and patients amidst a nationwide shortage of Rho(D) immune globulin therapy (RhIg)
- In April 2024, BillionToOne introduced the UNITY Fetal RhD NIPT as a dependable and convenient alternative to traditional methods for determining fetal RhD status, showcasing diagnostic-level accuracy with >99.9% sensitivity and specificity as well as 100% concordance with newborn outcomes, including the detection of challenging RhD Psi and RhD-CE, as demonstrated in recent data published in Scientific Reports
- In November 2023, Yourgene Health and Laboriad introduced non-invasive prenatal testing (NIPT) to Morocco, with Yourgene Health installing the initial NIPT platform, the IONA test, at the Centre de Biologie Riad (Laboriad), empowering Laboriad to furnish expectant parents with safe, rapid, and precise prenatal testing locally, alleviating the necessity for invasive procedures and alleviating the stress and anxiety often experienced by expectant parents awaiting results
- In April 2023, Mediclinic unveiled safe, non-invasive prenatal testing (NIPT) for expectant parents as part of their DNA-based diagnostic and clinical interpretation services, encompassing screening for common chromosomal conditions in the fetus, including Down syndrome, Patau syndrome, Edwards syndrome, sex chromosome aneuploidies, and optional screening for up to five microdeletion syndromes that could be overlooked during routine prenatal screening
The non-invasive prenatal testing market is expected to gain further momentum in the coming years due to rising incidences of genetic disorders, growing demand for non-invasive testing procedures, technological advancements, new product launches, and aggressive organic and inorganic growth strategies followed by the players.
| Report Scope | Details |
| Base Year Considered | 2023 |
| Historical Data | 2022 - 2023 |
| Forecast Period | 2024 - 2029 |
| CAGR (2024 - 2029) | 16% |
| Segment Scope | Product, Application, End User |
| Regional Scope |
|
| Key Companies Mapped | Agilent Technologies, Inc., PerkinElmer, Inc., Illumina, Inc., Eurofins LifeCodexx AG, Natera, Inc., Igenomix SL, F. Hoffman-La Roche Ltd., BGI Genomics, Cradle Genomics, Quest Diagnostics, Progenity, Inc., Berry Genomics, Sequenom, Roche (Ariosa Diagnostics), among others. |
| Report Highlights | Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis |
Key Strategic Questions Addressed
-
What is the market size & forecast for the Global Non-Invasive Prenatal Testing Market?
-
What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global Non-Invasive Prenatal Testing Market?
-
How has COVID-19 impacted the Global Non-Invasive Prenatal Testing Market?
-
What are the major growth drivers, restraints/challenges impacting the market?
-
What are the opportunities prevailing in the market?
-
What is the investment landscape?
-
Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
-
Who are the major players operating in the market? What is the competitive positioning of key players?
-
Who are the new players entering the market?
-
What are the key strategies adopted by players?
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Market Forecasting
- Executive Summary
- Market Overview
- Market Dynamics
- Drivers
- Restraints
- Key Market Trends
- Market Dynamics
-
- Industry Speaks
- Key Revenue Pockets
- Global Non-Invasive Prenatal Testing Market - Size & Forecast (2021-2028), By Product Type
- Consumables
- Kits & Reagents
- Instruments
- Others
- Global Non-Invasive Prenatal Testing Market - Size & Forecast (2021-2028), By Application Type
- Trisomy
- Microdeletion Syndrome
- Others
- Global Non-Invasive Prenatal Testing Market - Size & Forecast (2021-2028), By End User
- Hospitals
- Clinics
- Diagnostic Laboratories
- Others
- Global Non-Invasive Prenatal Testing - Size & Forecast (2021-2028), By Region
- North America (U.S. & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Rest of the World (Latin America, Middle East & Africa)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Competitive Positioning of Key Players (2022)
- Offerings Assessment, By Player
- Key Strategies Assessment, By Player (2021-2023)
- New Product & Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Key Players and their Competitive Positioning
- Key Companies Scanned (Indicative List)
- Agilent Technologies, Inc.
- PerkinElmer, Inc.
- Illumina, Inc.
- Eurofins LifeCodexx AG
- Natera, Inc.
- Igenomix SL
- Hoffman-La Roche Ltd.
- BGI Genomics
- Quest Diagnostics
- Berry Genomics
- Yourgene Health
- Other Prominent Players
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
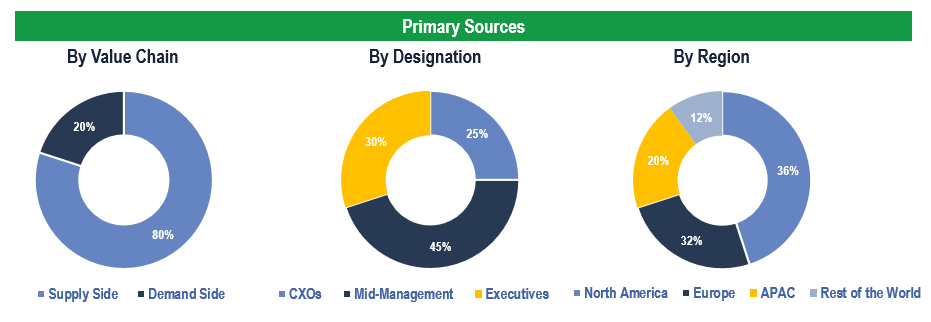
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Clinics, Diagnostic Laboratories and Other End Users
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Frequently Asked Questions (FAQs)
Features of the Report
- Comprehensive Market Coverage
- Market Size and Forecast
- Geographic & Segment Deep Dives
- Strategic Insights & Competitive Landscape
- Timely & Updated Data
- Growth Indicators & Future Outlook
- Quick Turnaround on Queries
- Analyst Support
- Report Customization Available
- Reports in PDF & Excel