navigating-the-preeclampsia-diagnostics-market-landscape-trends-innovations-and-challenges
Preeclampsia is a serious pregnancy-related condition characterized by elevated blood pressure and excess protein in the urine, posing significant risks to both mothers and infants globally. It often leads to premature and underweight births and is a prominent hypertensive disorder during pregnancy in developed and emerging nations. Failure to diagnose preeclampsia early can result in complications such as preterm birth, organ failure, seizures, eclampsia, and potentially fatal outcomes for both mother and baby. The only cure for preeclampsia at present is delivery, which significantly increases the difficulties associated with preterm births, neonatal morbidity, and perinatal mortality. Regular prenatal care and monitoring are essential for early detection and effective management of this condition.
Preeclampsia Diagnostics Global Market Dynamics
Preeclampsia is a pregnancy complication characterized by high blood pressure and signs of damage to organs, typically the liver and kidneys. The rising incidence of preeclampsia globally has been a significant driver for the development of new diagnostic and therapeutic solutions. Ongoing advancements in diagnostic technologies have led to improved and early detection of preeclampsia, facilitating timely intervention and management. For instance,
- In November 2022, Biora Therapeutics, Inc. disclosed the successful completion of a licensing agreement with Avero Diagnostics (formerly Northwest Pathology) for the commercial development of its Preecludia™ rule-out test for preeclampsia
Pharmaceutical companies and research institutions are actively involved in R&D efforts to develop effective and less invasive solutions for preeclampsia, which can further drive market growth. Increased awareness of preeclampsia and government initiatives, along with funding for research projects, contribute to the market's growth. Collaborations between pharmaceutical companies, academic institutions, and healthcare organizations are crucial in advancing research and developing new solutions for preeclampsia. Efforts to educate patients and healthcare professionals about the risks and early signs of preeclampsia contribute to early diagnosis and treatment, positively impacting the market.
Fill out the "Quick Inquiry Form" to request a sample copy
Preeclampsia: Risk Factors, Diagnosis, and the Quest for Predictive Biomarkers
Preeclampsia, a common condition in pregnancy is on the rise globally. The need for diagnostic tests for preeclampsia has escalated due to elevated blood pressure during pregnancy, attributed to lifestyle changes, inadequate diet, sedentary habits, alcohol consumption, and mental stress. The increasing occurrences of preeclampsia can be attributed to risk factors such as advanced maternal age, obesity, and diabetes.
For many years, physicians have commonly used blood pressure and urine protein measurements to assess the likelihood of a woman developing preeclampsia. However, these tests prove inadequate in predicting the development of severe preeclampsia, particularly as the pregnancy advances. Preeclampsia lacks a dedicated diagnostic test, and the sole curative measure involves delivering both the baby and the placenta, with the latter thought to be a contributing factor to the condition. In cases where the infant may not survive outside the womb, healthcare professionals employ a conservative approach to manage the mother's condition, utilizing antihypertensive medications, magnesium, and steroids.
Recently there has been a growing focus on predictive biomarkers for preeclampsia. A reliable predictive test simplifies early diagnosis, enables targeted surveillance, and facilitate timely delivery. A biomarker capable of identifying high-risk women in early pregnancy holds clinical significance in preventing preterm preeclampsia. For instance,
- In June 2023, Diabetomics presented the results of clinical studies on first-trimester screening and diagnostic support for preeclampsia at the 20th World Congress in Fetal Medicine in Valencia, Spain. The result stated that the addition of GlyFn as a supplementary biomarker to the FMF 'Triple' screening test for preterm preeclampsia has notably improved the screening performance, but further validation in diverse populations is necessary to confirm these findings
- In May 2023, Thermo Fisher's received FDA approval for its immunoassays designed for the evaluation of preeclampsia risk. These are the first immunoassays to receive breakthrough designation and FDA clearance, which will assess the risk of developing preeclampsia and support clinical management, marking a significant advancement in addressing this severe pregnancy complication
Key Constraints/Challenges
The complex nature of preeclampsia necessitates a multidisciplinary approach, involving specialists in maternal-fetal medicine, obstetricians, and various healthcare professionals. The integration of such services may encounter barriers, posing challenges to market growth. The lack of awareness about preeclampsia in developing countries along with inadequate awareness among healthcare providers about effective screening tools for the condition is expected to hinder market expansion in emerging countries.
North America Accounts for the Largest Share of the Global Market
The North America region is leading the global market for preeclampsia diagnostics, holding the largest market share. This dominance is attributed to the rising healthcare expenditure, widespread adoption of advanced products, and favourable initiatives by the government, acknowledging the severity of the condition. The growing prevalence of preeclampsia is resulting in escalated medical treatment costs for women affected by preeclampsia and their newborns. The Asia-Pacific region is anticipated to grow at the highest CAGR, mainly attributed to the high pregnancy rate, rising awareness about the disease and the increasing population in the region.
Competitive Landscape
The global preeclampsia diagnostics market is dominated by some of the key players which include Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd, PerkinElmer Inc., Siemens Healthineers AG, DRG Instruments GMBH, Metabolomic Diagnostics Ltd., Bayer AG, Sera Prognostics, and Diabetomics, Inc. among others.
Organic and Inorganic Growth Strategies Adopted by Players to Establish Their Foothold in the Market
Players operating in this market are adopting both organic and inorganic growth strategies such as receiving grants for research, entering into collaborations, and acquiring related firms to garner market share. For instance,
- In February 2023, Boston University's Wendy Kuohung, MD, received a grant of $99,994 USD from the Preeclampsia Foundation for new research aimed at investigating genetic targets with the potential to develop therapeutics/diagnostic solutions for preeclampsia.
- In May 2022, Sera Prognostics Inc., recognized as The Pregnancy Company®, in collaboration with the Newborn Foundation, unveiled the initiation of the Every Mother, Every Baby Project. This initiative aims to reduce maternal and neonatal mortality and morbidity by addressing risks linked to unexpected preterm birth.
The market is expected to grow due to the rising prevalence of preeclampsia, increasing R&D initiatives for preeclampsia biomarkers, growing demand for advanced diagnostic tests, and aggressive organic and inorganic growth strategies followed by the players.
Preeclampsia Diagnostics Market Scope
|
Report Metric
|
Details
|
|
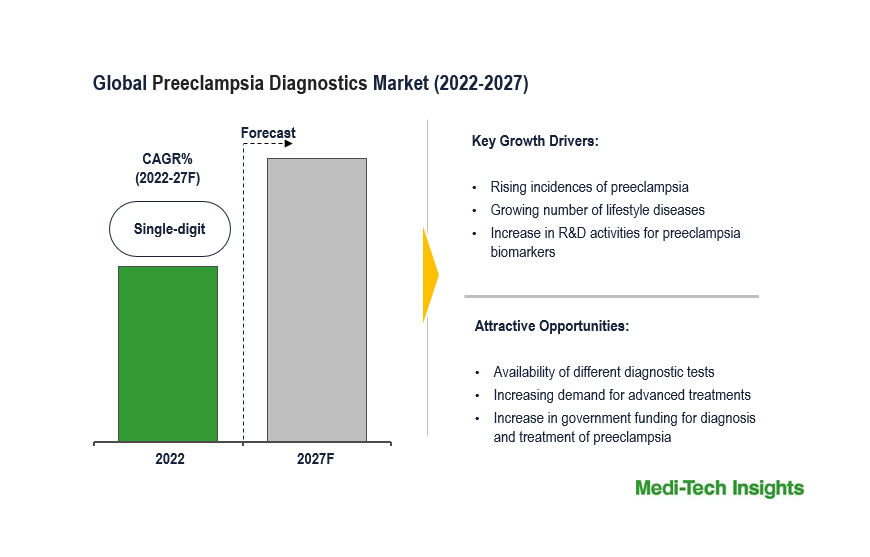
Market Growth Rate (2027)
|
Single-digit growth rate is expected.
|
|
Drivers of Market Growth
|
- Increasing prevalence of preterm births.
- Rising number of lifestyle diseases.
- Growing demand for advanced diagnostics tests.
- Surge in R&D activities for preeclampsia biomarkers.
|
|
Risk Factors for Preeclampsia
|
Advanced maternal age, obesity, and diabetes. Lifestyle factors include inadequate diet, sedentary habits, alcohol consumption, and mental stress.
|
|
Diagnostic Methods
|
Blood pressure and urine protein measurements. Lack of dedicated diagnostic test. Focus on predictive biomarkers for early diagnosis and timely delivery.
|
|
Key Players in the Market
|
Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd, PerkinElmer Inc., Siemens Healthineers AG, DRG Instruments GMBH, Metabolomic Diagnostics Ltd., Bayer AG, Sera Prognostics, Diabetomics, Inc., among others.
|
|
Future Market Outlook
|
- Expected growth due to rising preeclampsia prevalence.
- Increasing R&D initiatives for biomarkers.
- Growing demand for advanced diagnostics.
- Aggressive organic and inorganic growth strategies by market players.
|
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)
Primary Research
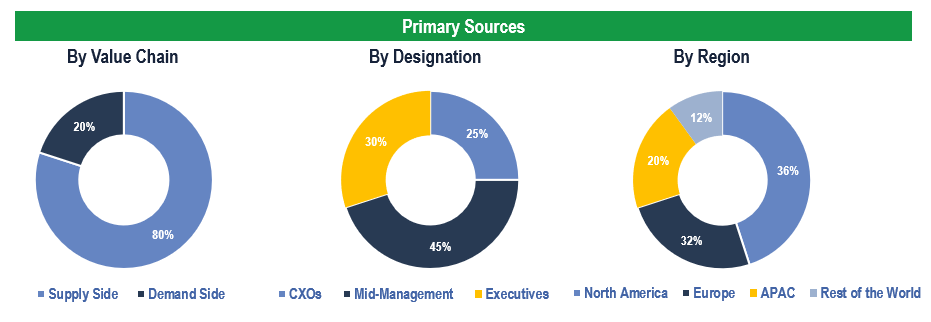
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Stakeholders in Hospitals, Specialty Clinics, Diagnostics Centers and Other End Users
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down and Bottom-Up Approaches’ were used to derive market size estimates and forecasts.
Data Triangulation
Research findings derived through secondary sources & internal analysis were validated with Primary Interviews, Internal Knowledge Repository, and Company Sales Data.
Key Strategic Questions Addressed
- What is the market size & forecast for the Global Preeclampsia Diagnostics Market?
- What are the historical, present, and forecasted market shares and growth rates of various segments and sub-segments of the Global Preeclampsia Diagnostics Market?
- How has COVID-19 impacted the Global Preeclampsia Diagnostics Market?
- What are the major growth drivers, restraints/challenges impacting the market?
- What are the opportunities prevailing in the market?
- What is the investment landscape?
- Which region has the highest share in the global market? Which region is expected to witness the highest growth rate in the next 5 years?
- Who are the major players operating in the market? What is the competitive positioning of key players?
- Who are the new players entering the market?
- What are the key strategies adopted by players?