lipid-nanoparticle-cdmo-market-size-share-growth-trends-and-competitive-analysis-report-2027
LNPs are nanosized particles primarily composed of lipids, utilized as drug delivery systems due to their biocompatibility and ability to encapsulate various therapeutic agents. They play a crucial role in modern medicine, particularly in the delivery of mRNA vaccines and other therapeutics. LNP CDMO is a type of service provider that specializes in assisting with the development, formulation, and large-scale commercial production of LNP drug delivery systems for both pharmaceutical and biotech companies. LNP CDMO's provide full-service solutions which include, process development, analytical testing, formulation optimization, and good manufacturing practices (GMP) compliant production of LNPs utilized in mRNA, siRNA, and other nucleic acid therapeutic modalities. LNP CDMOs are a key contributor to shorten the drug development timeframe; enabling the scalable and reproducible manufacturing of products; and meeting regulatory compliance requirements due to an increasing number of novel therapies including vaccines and gene-based therapy development in numerous areas of therapeutics.
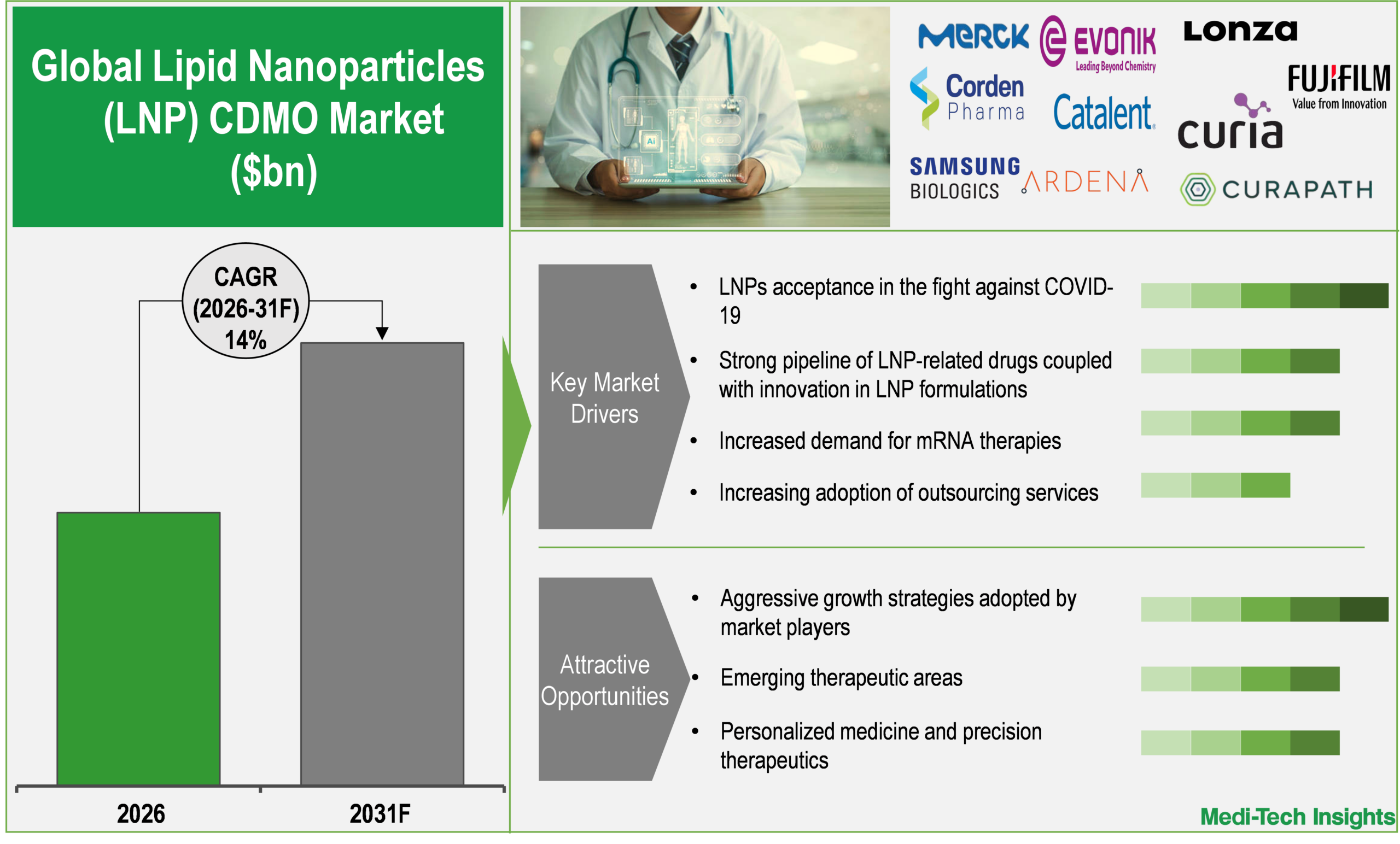
Strong pipeline of LNP-related drugs coupled with innovation in LNP formulations to propel market demand
There is a consistent demand for LNPs because of a growing number of LNP-based therapeutics and LNP formulation innovations. The use of LNPs as an effective delivery system has been validated using mRNA vaccines during the COVID-19 pandemic that spurred the growth and expansion of pharmaceutical and biotechnology companies' LNP-enabled drug pipelines. As of 2025, there were over 150 RNA-LNP formulations in clinical trials, driving a significant demand for specialized services from CDMOs (Source: NCBI). LNP-enabled drug pipelines now have various applications that are being pursued, including vaccines, gene therapies, cancer immunotherapies and treatments for rare genetic disorders. As more drug candidates progress through the continuum of drug development from preclinical to clinical and commercial stages, the demand for CDMOs with expertise in developing specialized products is likely to increase and directly benefit LNP CDMOs.
The diversification and complexity of drug candidates based on LNPs is another factor contributing to the increasing demand for CDMOs with specialized capabilities in developing and manufacturing LNPs. These customized LNP formulations are based on how each therapeutic application is designed to achieve optimized delivery, stability, bio-distribution, and safety. For instance, the distinct types of LNPs are required for the targeted delivery of siRNA to the liver compared to systemic mRNA vaccines versus localized cancer therapies. The need for customized formulation development for each drug candidate could lead to an increase in the development of partnerships between pharmaceutical companies and CDMOs with advanced expertise in lipid chemistry, nanoparticle engineering and scalability of manufacturing processes.
An additional key driver of growth within LNPs is innovations in LNP formulation technology. Innovative technologies such as ionizable lipids, advanced encapsulation techniques, and advanced targeted delivery mechanisms are improving the performance of LNP-based therapeutics and the likelihood of clinical success of those therapeutics. Moreover, several new formulation strategies are being developed to meet the challenges related to the storage stability of LNPs, the cold-chain requirements of handling LNPs, and repeat dosing/congruencies with LNPs in order to make these therapies commercially viable.
On the other hand, developing and commercializing advanced LNP formulations requires sophisticated infrastructure, proprietary technology, and a highly specialized workforce that many pharmaceutical companies do not have within their organizations. Consequently, outsourcing to LNP-focused CDMOs is strategic necessity for the pharmaceutical industry. In addition to providing technical capability, CDMOs can assist in obtaining regulatory approval for LNP-based therapeutics and can accelerate time-to-market for those therapeutics.
In addition, many LNP-focused CDMOs are investing in the development of proprietary LNP platforms and expanding their service offerings to differentiate themselves from one another, in response to the competitive nature of the LNP CDMO marketplace. This creates a positive cycle whereby innovation is driving pipeline expansion, and increases in the pipelines drive outsourcing demand, ultimately leading to continued long-term growth in the LNP CDMO market.
To learn more about this report, download the PDF brochure
Increasing adoption of outsourcing services is driving the market growth
Rising outsourcing is a significant driver of the growth of the LNP CDMO market, as pharmaceutical and biotech companies look for alternatives to carry out LNP based therapeutic development and manufacturing in house due to technical, operational, and financial challenges inherent in doing that. Many companies, especially small and mid-sized biotech businesses do not have the necessary expertise in lipid chemistry, nanoparticle engineering or encapsulation technologies to develop LNP formulations, particularly for mRNA, siRNA, and gene therapies, so outsourcing is often necessary due to their lack of infrastructure and skilled personnel to manage these types of complex processes internally.
Outsourcing to an LNP focused CDMO enables companies to gain access to advanced technologies without making expensive capital expenditures to establish LNP manufacturing within their own facilities. Developing an in-house LNP manufacture facility involves high costs related to specialized equipment, cleanroom environments, and compliance with stringent regulatory standards such as GMP. By partnering with CDMOs, companies can benefit from the technical and infrastructural advantages of working with an experienced and well-established manufacturer, allowing the company to apply their resources more effectively toward their expertise in the core areas of research and clinical development.
The use of outsourcing adds flexibility, convenience, and scalability during various points of the drug development process. As the LNP-based candidate advances through research to clinical trials to final market access, the production needs often change significantly. CDMOs provide scalable manufacturing options that can accommodate changing batch sizes and timelines, allowing for seamless movement from one phase of development to another. This flexibility is especially important among the dynamic changes occurring within the therapeutic landscape, where the need for speed and responsiveness is paramount.
Another key factor driving the use of CDMOs is their ability to expedite the time it takes for products to reach the market. Due to their well-established procedures, knowledgeable team members, and regulatory expertise; CDMOs can streamline timelines and mitigate delays related to optimizing the manufacturing process and navigating through the regulatory approval process. This is of significant value in rapidly growing and competitive therapeutic landscapes like mRNA-based products, where the first company to commercialize a product is likely to have a clear advantage over its competitors.
The continued rise in demand for LNP-based products has led to an increase in the trend toward long-term outsourcing or strategic partner agreements that reflect a growing confidence in the role of CDMOs as integral players in drug development. These factors may likely sustain the use of outsourcing as a strategic business model for LNP-based therapies and subsequently, the continued growth and innovation within the LNP CDMO market.
To learn more about this report, download the PDF brochure
Phase Segment Outlook
The commercial phase including large-scale cGMP manufacturing, process optimization, fill & finish, supply chain management, regulatory support, and quality control is estimated to be the largest revenue-generating segment for LNP CDMOs, largely due to ongoing production of the innovative vaccines and other approved LNP therapies. The clinical phase includes services such as process optimization, phase-specific method validation, clinical-scale cGMP manufacturing, regulatory support, and clinical trial supply management, among others, is the second largest segment owing to clinical trials of various RNA-based therapeutics for different therapeutic applications. The clinical phase is expected to be the fastest-growing segment during the forecast period. The growth in this segment is largely attributed to factors like growing demand for advanced RNA therapies, increasing number of clinical trials, and rising demand for high-quality, consistent clinical supplies.
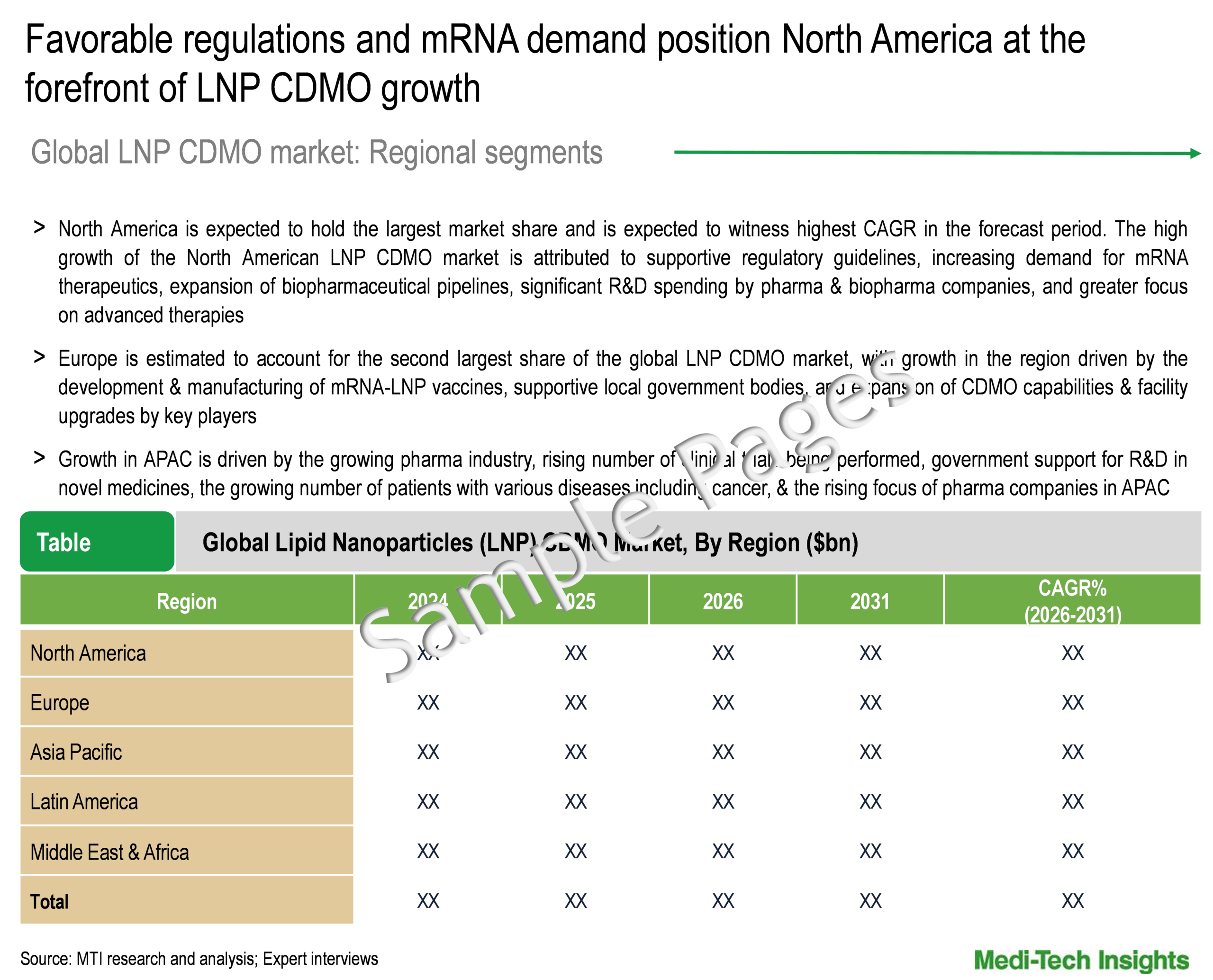
Regional Outlook: Favourable Regulations and Rising mRNA Demand Position North America at the Forefront of LNP CDMO Market
North America is expected to hold the largest market share and is expected to witness highest CAGR in the forecast period. The high growth of the North American LNP CDMO market is attributed to supportive regulatory guidelines, increasing demand for mRNA therapeutics, expansion of biopharmaceutical pipelines, significant R&D spending by pharma & biopharma companies, and greater focus on advanced therapies. Furthermore, support from governments, especially in the US, for advanced biomanufacturing and the development of next-generation therapies is expected to boost the LNP CDMO market during the forecast period. Regulatory approvals and fast-track designations for LNP-based drugs further enhance market prospects. Europe is estimated to account for the second largest share of the global LNP CDMO market, with growth in the region driven by the development & manufacturing of mRNA-LNP vaccines, supportive local government bodies, and expansion of CDMO capabilities & facility upgrades by key players. Growth in APAC is driven by the growing pharma industry, rising number of clinical trials being performed, government support for R&D in novel medicines, growing number of patients with various diseases including cancer, and the rising focus of pharma companies in the region.
Competitive Landscape Analysis
The global LNP CDMO market is marked by the presence of established and emerging market players such as Merck KGaA (Germany); Evonik Industries AG (Germany); Corden Pharma (Switzerland); Catalent Inc. (US); Lonza Group (Switzerland); FUJIFILM Pharmaceuticals U.S.A., Inc. (US); Samsung Biologics Co., Ltd. (S. Korea); Curia Global, Inc. (USA); Ardena Holdings (Belgium); and Curapath (US); among others.
Get a sample report for competitive landscape analysis
Growth strategies adopted by players to establish their foothold in the market
Players operating in this market are adopting various growth strategies such as new service launches, and strategic partnerships, agreements, and collaborations to garner market share. For instance,
- In September 2025, Evonik Industries and Ethris entered a strategic partnership to develop and commercialize an advanced LNP platform for nucleic acid delivery, particularly in respiratory therapies. The collaboration enhances CDMO capabilities by combining proprietary SNaP LNP technology with large-scale manufacturing expertise, enabling more stable, targeted, and scalable RNA-based therapeutics
- In September 2025, Porton Advanced and RongCan Biotech announced a strategic collaboration to co-develop and commercialize novel LNP delivery technologies for nucleic acid and cell therapies. The partnership combines innovative ionizable lipid platforms with large-scale CDMO capabilities, enabling integrated development, licensing, and manufacturing to accelerate advanced therapy commercialization
- In February 2024, Corden Pharma International launched LNP starter kits to support mRNA therapeutic development by providing essential lipids for formulation and optimization. The kits enable efficient screening and scalable LNP development, helping researchers accelerate mRNA drug development and ensure consistency from R&D to clinical stages.
Report Scope
|
Report Metric
|
Details
|
|
Base Year Considered
|
2025
|
|
Historical Data
|
2024 – 2025
|
|
Forecast Period
|
2026 – 2031
|
|
Growth Rate
|
14%
|
|
Segment Scope
|
Phase, Application, End User
|
|
Regional Scope
|
- North America (US & Canada)
- Europe (UK, Germany, France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, India, Japan, Rest of Asia Pacific)
- Latin America
- Middle East & Africa
|
|
Market Drivers
|
- Strong pipeline of LNP-related drugs coupled with innovation in LNP formulations
- Increased demand for mRNA therapies
- Increasing adoption of outsourcing services
- Growing demand of LNP for infectious diseases and other medical applications
- LNPs acceptance in the fight against COVID-19
- A growing trend of developing proprietary LNP platforms among CDMOs
|
|
Attractive Opportunities
|
- Aggressive growth strategies adopted by market players
- Emerging therapeutic areas
- Personalized medicine and precision therapeutics
- Small and mid-sized pharmaceutical and biotech companies
|
|
Key Companies Mapped
|
Merck KGaA (Germany); Evonik Industries AG (Germany); Corden Pharma (Switzerland); Catalent Inc. (US); Lonza Group (Switzerland); FUJIFILM Pharmaceuticals U.S.A., Inc. (US); Samsung Biologics Co., Ltd. (S. Korea); Curia Global, Inc. (USA); Ardena Holdings (Belgium); and Curapath (US); among others
|
|
Report Highlights
|
Market Size & Forecast, Growth Drivers & Restraints, Trends, Competitive Analysis
|
Global Lipid Nanoparticles CDMO Market Segmentation
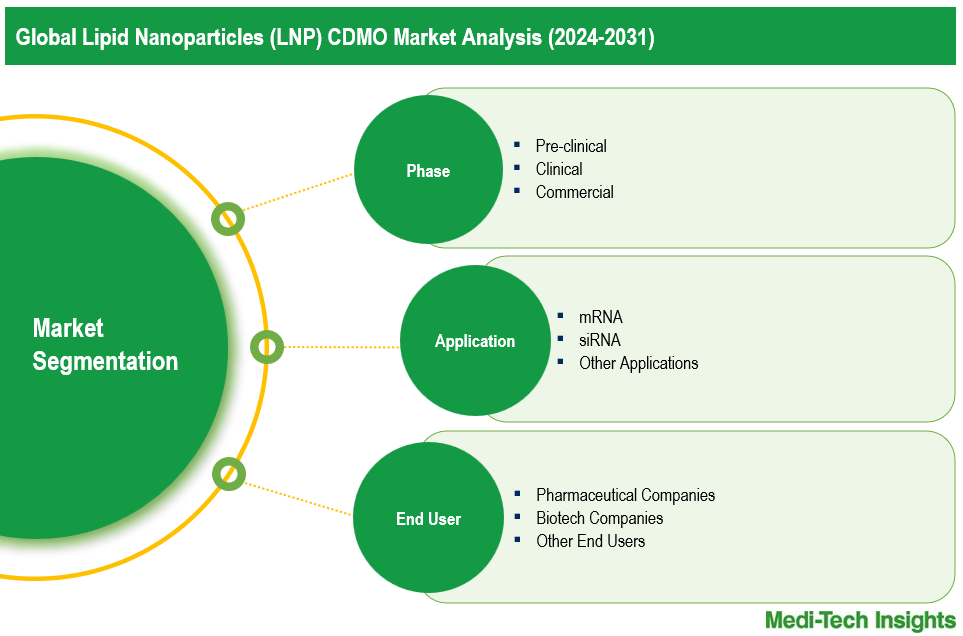
This report by Medi-Tech Insights provides the size of the global LNP CDMO market at the regional- and country-level from 2024 to 2031. The report further segments the market based on phase, application, and end user.
Market Size & Forecast (2024-2031), By Phase, USD Billion
- Pre-clinical
- Clinical
- Commercial
Market Size & Forecast (2024-2031), By Application, USD Billion
- mRNA
- siRNA
- Other Applications
Market Size & Forecast (2024-2031), By End User, USD Billion
- Pharmaceutical Companies
- Biotech Companies
- Other End Users
Market Size & Forecast (2024-2031), By Region, USD Billion
- North America
- Europe
- UK
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Middle East & Africa
- Introduction
- Introduction
- Market Scope
- Market Definition
- Segments Covered
- Regional Segmentation
- Research Timeframe
- Currency Considered
- Study Limitations
- Stakeholders
- List of Abbreviations
- Key Conferences and Events (2026-2027)
- Research Methodology
- Secondary Research
- Primary Research
- Market Estimation
- Bottom-Up Approach
- Top-Down Approach
- Market Forecasting
- Executive Summary
- Lipid Nanoparticles (LNP) CDMO Market Snapshot (2026-2031)
- Segment Overview
- Regional Snapshot
- Competitive Insights
- Market Overview
- Market Dynamics
- Drivers
- Strong pipeline of LNP-related drugs coupled with innovation in LNP formulations
- Increased demand for mRNA therapies
- Increasing adoption of outsourcing services
- LNPs acceptance in the fight against COVID-19
- Growing demand of LNP for infectious diseases and other medical applications
- A growing trend of developing proprietary LNP platforms among CDMOs
- Restraints
- Regulatory hurdles
- Supply chain inconsistencies
- Complex manufacturing processes
- Opportunities
- Aggressive growth strategies adopted by market players
- Emerging therapeutic areas
- Personalized medicine and precision therapeutics
- Small and mid-sized pharmaceutical and biotech companies
- Key Market Trends
- Growing investments by private equity companies in lipid nanoparticle CDMOs
- Expansion of CDMO capabilities
- Continuous manufacturing
- Single-use technologies (SUTs)
- Unmet Market Needs
- Industry Speaks
- Global Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Phase, USD Billion
- Introduction
- Pre-clinical
- Clinical
- Commercial
- Global Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Application, USD Billion
- Introduction
- mRNA
- siRNA
- Other Applications
- Global Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By End User, USD Billion
- Introduction
- Pharmaceutical Companies
- Biotech Companies
- Other End Users
- Global Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Region, USD Billion
- Introduction
- North America Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Country, USD Billion
- US
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Canada
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Europe Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Country, USD Billion
- UK
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Germany
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- France
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Italy
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Spain
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Rest of Europe
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Asia Pacific (APAC) Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), By Country, USD Billion
- China
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Japan
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- India
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Rest of Asia Pacific
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Latin America (LATAM) Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), USD Billion
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Middle East & Africa (MEA) Lipid Nanoparticles (LNP) CDMO Market Size & Forecast (2024-2031), USD Billion
- Market Size & Forecast, By Phase, (USD Billion)
- Market Size & Forecast, By Application, (USD Billion)
- Market Size & Forecast, By End User, (USD Billion)
- Competitive Landscape
- Key Players and their Competitive Positioning
- Key Player Comparison
- Segment-wise Player Mapping
- Market Share Analysis (2025)
- Company Categorization Matrix
- Dominants/Leaders
- New Entrants
- Emerging Players
- Innovative Players
- Key Strategies Assessment, By Player (2023-2026)
- New Service Launches
- Partnerships, Agreements, & Collaborations
- Mergers & Acquisitions
- Geographic Expansion
- Company Profiles*
(Business Overview, Financial Performance**, Products Offered, Recent Developments)
- Merck KGaA
- Evonik Industries AG
- Corden Pharma
- Catalent Inc.
- Lonza Group
- FUJIFILM Pharmaceuticals U.S.A., Inc.
- Samsung Biologics Co, Ltd.
- Curia Global, Inc.
- Ardena Holdings
- Curapath
- Other Prominent Players
Note: *Indicative list
**For listed companies
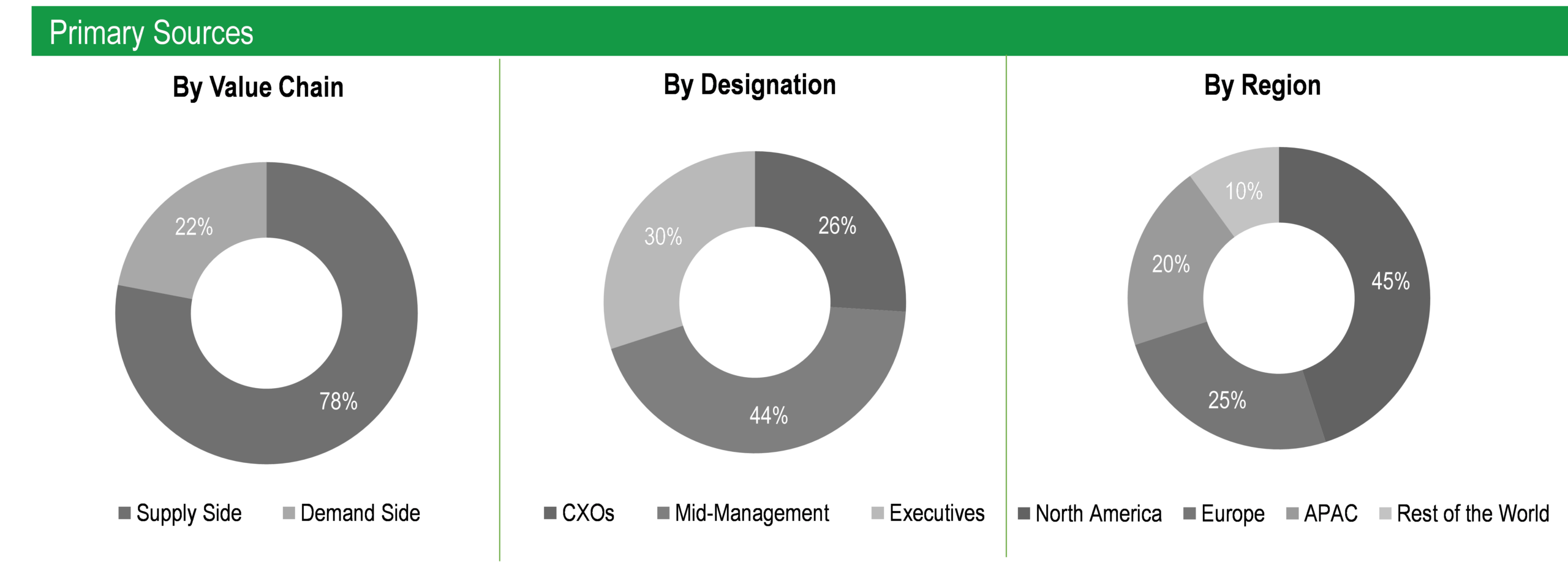
The study has been compiled based on extensive primary and secondary research.
Secondary Research (Indicative List)

Primary Research
To validate research findings (market size & forecasts, market segmentation, market dynamics, competitive landscape, key industry trends, etc.), extensive primary interviews were conducted with both supply and demand-side stakeholders.
Supply Side Stakeholders:
- Senior Management Level: CEOs, Presidents, Vice-Presidents, Directors, Chief Technology Officers, Chief Commercial Officers
- Mid-Management Level: Product Managers, Sales Managers, Brand Managers, R&D Managers, Business Development Managers, Consultants
Demand Side Stakeholders:
- Pharmaceutical Companies, Biotech Companies, and Others
Breakdown of Primary Interviews
Market Size Estimation
Both ‘Top-Down & Bottom-Up Approaches’ were used to derive market size estimates and forecasts
Data Triangulation
Research findings derived through secondary sources & internal analysis was validated with Primary Interviews, Internal Knowledge Repository and Company’s Sales Data